Research Articles
Bridging the Predictive Gap: Advances in Correlating In Vitro and In Vivo Toxicity for Modern Drug Development
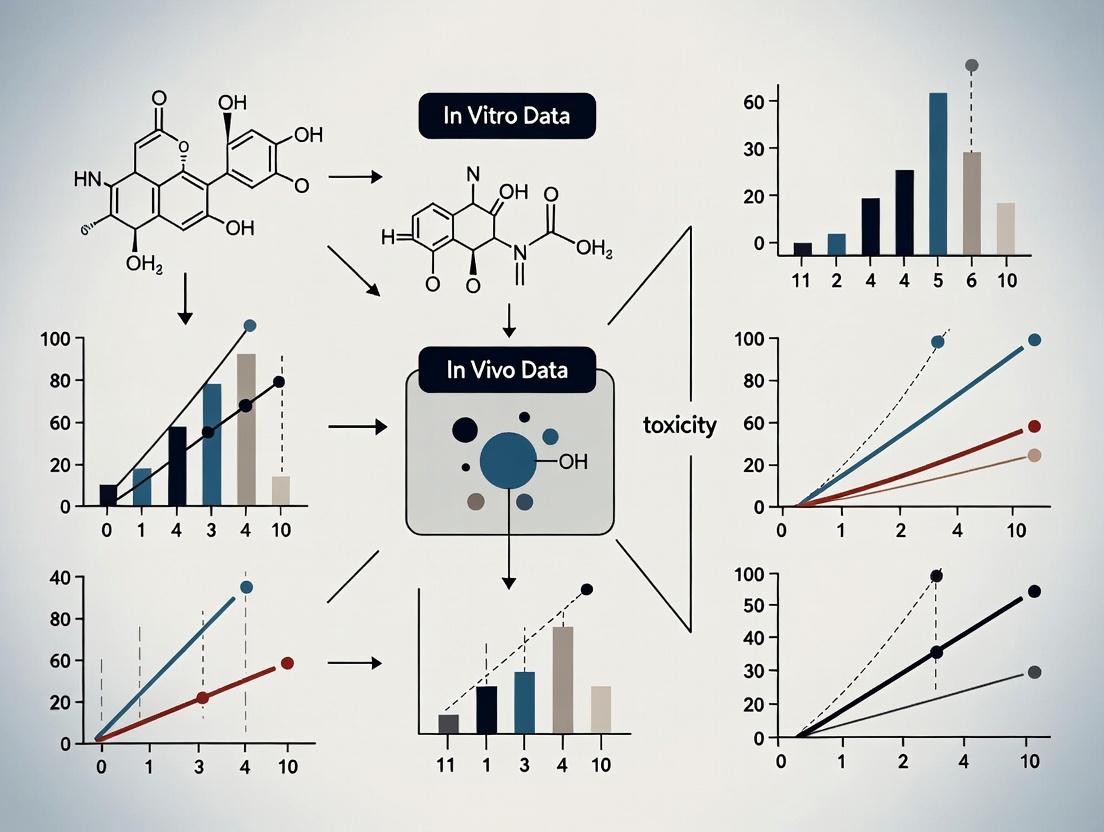
This article provides a comprehensive examination of the correlation between in vitro and in vivo toxicity data, a critical nexus in pharmaceutical and chemical safety assessment.
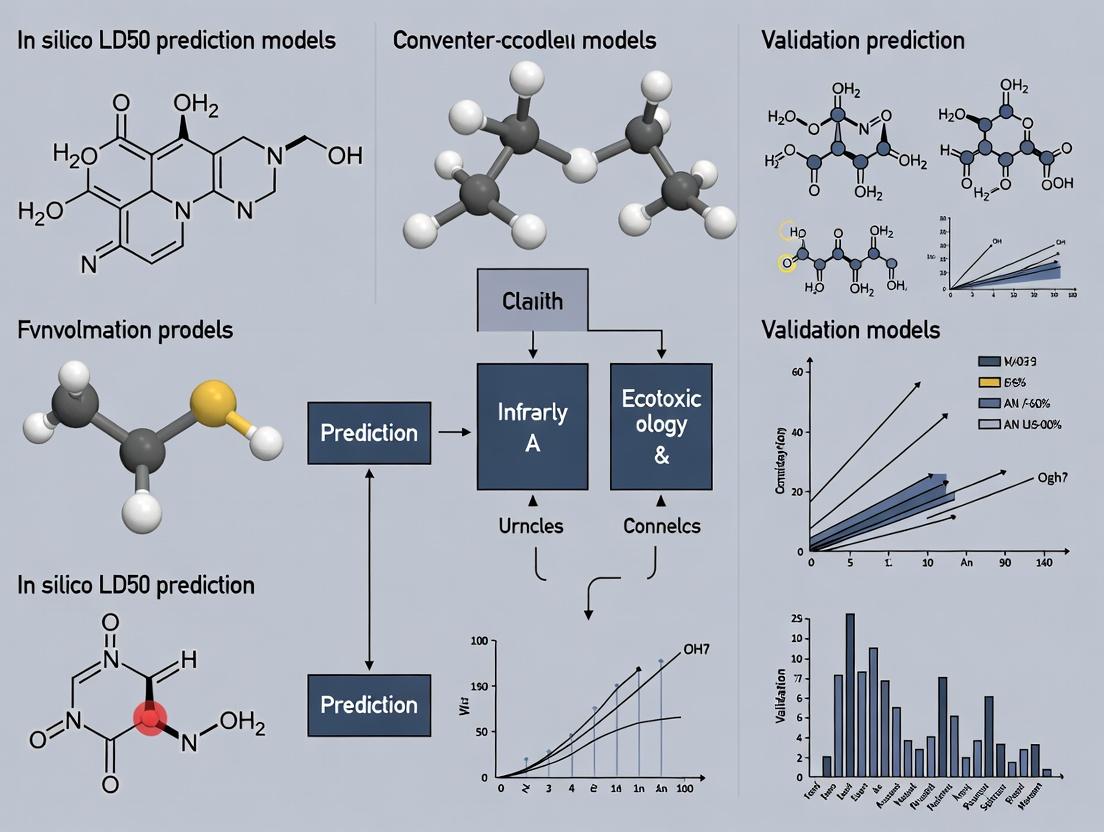
Validating In Silico LD50 Models: A Comprehensive Guide for AI-Driven Toxicity Prediction in Drug Discovery
This article provides a systematic framework for researchers and drug development professionals to evaluate and validate in silico models for predicting acute oral toxicity (LD50).
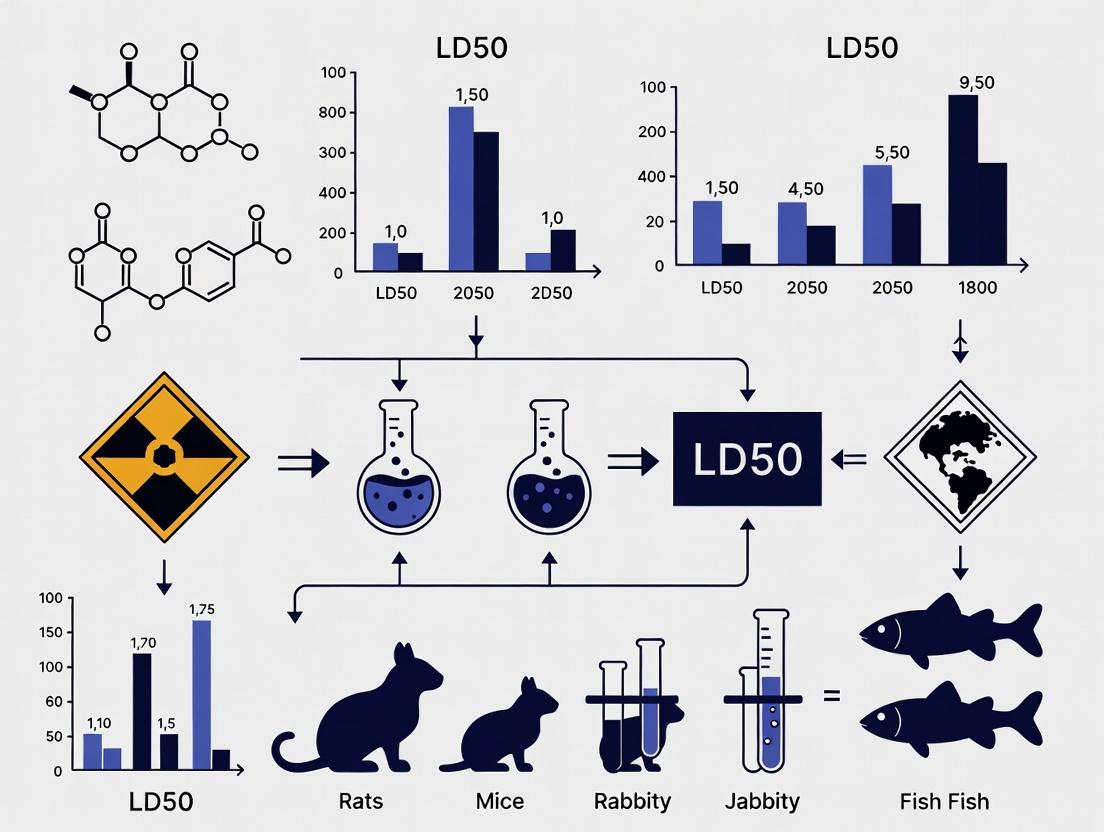
Beyond Rodent Models: A Systematic Guide to Cross-Species LD50 Comparison for Predictive Toxicology and Risk Assessment
This article provides a comprehensive analysis of methodologies and challenges in comparing Lethal Dose 50 (LD50) values across different species—a critical task for researchers, toxicologists, and drug development professionals.
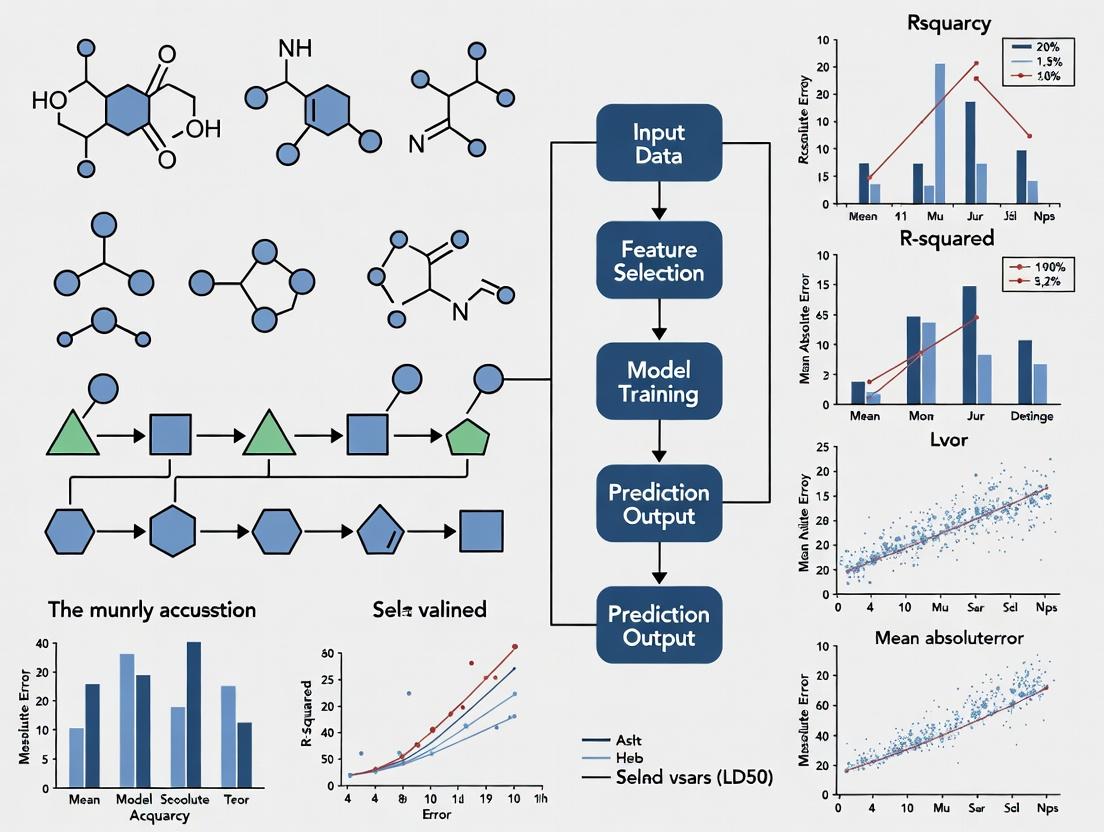
Beyond Animal Testing: How Machine Learning Models Achieve High-Fidelity LD50 Prediction for Health-Protective Risk Assessment
This article provides a comprehensive review for researchers and drug development professionals on the application of machine learning (ML) for predicting rat acute oral LD50, a critical parameter for chemical...
When NOAEL Cannot Be Determined: Modern Strategies and Alternative Approaches for Drug Safety Assessment
This article addresses the critical challenge in preclinical development when a No-Observed-Adverse-Effect Level (NOAEL) cannot be reliably determined.
Interlaboratory Variability in Toxicity Testing: Foundational Insights and Methodological Solutions for Drug Development
Interlaboratory variability in toxicity testing poses significant challenges for drug development, regulatory decisions, and clinical translations.
Small Sample Size Toxicity Studies: Statistical Methods, Challenges, and Solutions for Robust Research
This article provides a comprehensive overview of statistical methods for toxicity studies with small sample sizes, common in preclinical research.
Ensuring Accuracy and Reliability: A Comprehensive Guide to Quality Control in LD50 Laboratory Testing
This article provides a detailed examination of quality control in LD50 laboratory testing for researchers, scientists, and drug development professionals.
Bridging the Translational Gap: A Modern Guide to Species Selection & Human Relevance in Toxicology
This article provides a comprehensive guide for researchers and drug development professionals on navigating species differences in toxicity testing.
Improving LD50 Reproducibility: Strategies for Robust and Reliable Acute Toxicity Testing
This article provides a comprehensive framework for researchers and drug development professionals aiming to enhance the reproducibility of LD50 determinations.