Inside the ECOTOX Knowledgebase: A Guide to the Systematic Data Curation Process for Chemical Risk Assessment
This article provides a comprehensive guide to the data curation process of the ECOTOX Knowledgebase, the world's largest compilation of curated ecotoxicity data.
Inside the ECOTOX Knowledgebase: A Guide to the Systematic Data Curation Process for Chemical Risk Assessment
Abstract
This article provides a comprehensive guide to the data curation process of the ECOTOX Knowledgebase, the world's largest compilation of curated ecotoxicity data. We detail the systematic, multi-stage pipeline—from literature search to final entry—that transforms raw scientific studies into a reliable, FAIR-compliant resource. Aimed at researchers, scientists, and drug development professionals, this guide explores ECOTOX's foundational role in regulatory science, offers practical methodologies for data extraction and application, addresses common challenges in data evaluation, and validates its use through real-world examples in chemical safety assessment and New Approach Methodologies (NAMs).
Understanding ECOTOX: The Foundation of Curated Ecotoxicity Data for Regulatory Science
What is the ECOTOX Knowledgebase? Defining the World's Largest Curated Ecotoxicity Resource
The exponential growth of chemicals in commerce necessitates efficient, reliable methods for ecological hazard assessment. In response, the U.S. Environmental Protection Agency (USEPA) developed the ECOTOXicology Knowledgebase (ECOTOX). Initiated in the 1980s and continuously refined, ECOTOX has evolved into the world's largest, publicly available compilation of curated single-chemical ecotoxicity data. It serves as a critical resource for regulators, researchers, and risk assessors, supporting chemical safety evaluations, ecological research, and the development of New Approach Methodologies (NAMs). This whitepaper provides a technical deep-dive into ECOTOX, framing its significance within a broader thesis on systematic data curation processes in environmental toxicology.
ECOTOX is a living database, updated quarterly with new data curated from the scientific literature. Its scale is a direct result of decades of systematic review and data abstraction. The following table summarizes the current quantitative scope of the knowledgebase as of its latest release.
Table 1: Quantitative Scope of the ECOTOX Knowledgebase (Version 5)
| Metric | Value | Source |
|---|---|---|
| Compiled References | Over 53,000 peer-reviewed and grey literature sources | [reference:0] |
| Total Test Records | Over 1 million curated toxicity test results | [reference:1] |
| Unique Chemicals | Approximately 12,000 single chemical stressors | [reference:2] |
| Ecological Species | More than 13,000 aquatic and terrestrial species | [reference:3] |
| Data Fields per Record | Over 100 structured fields for search and export | [reference:4] |
The Data Curation Process: A Thesis Perspective
The integrity and utility of ECOTOX are rooted in its transparent, standardized curation pipeline. This process aligns with contemporary systematic review practices and FAIR (Findable, Accessible, Interoperable, Reusable) data principles[reference:5]. The workflow is governed by detailed Standard Operating Procedures (SOPs) covering literature search, data abstraction, and maintenance[reference:6].
The core of the curation logic is a PECO (Population, Exposure, Comparator, Outcome) framework, which defines strict inclusion criteria for studies[reference:7].
- Population (P): Ecologically relevant, whole organisms (aquatic and terrestrial). Bacteria, viruses, and humans are excluded.
- Exposure (E): Single-chemical exposure with a verifiable CAS number, reported concentration, and duration. Air pollution studies (CO₂, ozone) are excluded.
- Comparator (C): A documented control treatment (e.g., vehicle-only) is required.
- Outcome (O): Measured biological effects concurrent with exposure.
Experimental Protocols: Standard Operating Procedures for Data Curation
The ECOTOX team's methodology for transforming primary literature into structured data can be considered a meta-experimental protocol. The key phases are:
1. Literature Search & Citation Identification: Comprehensive searches are conducted across open and grey literature databases using chemical-specific terms. Retrieved citations are initially screened by title and abstract[reference:8].
2. Applicability & Acceptability Screening: Full-text articles are reviewed against the PECO criteria. Studies must report essential details like chemical purity, species verification, test method (e.g., OECD guidelines), and appropriate controls to be deemed acceptable for data extraction[reference:9][reference:10].
3. Data Abstraction: For each accepted study, trained reviewers extract detailed information into over 100 structured fields. This includes chemical properties, species taxonomy, test conditions (media, duration, temperature), and quantitative results (e.g., LC50, NOEC, effect measurements)[reference:11].
4. Quality Control & Data Maintenance: Extracted data undergo rigorous quality checks. The underlying controlled vocabularies and SOPs are reviewed and updated quarterly to incorporate new efficiencies and maintain consistency[reference:12].
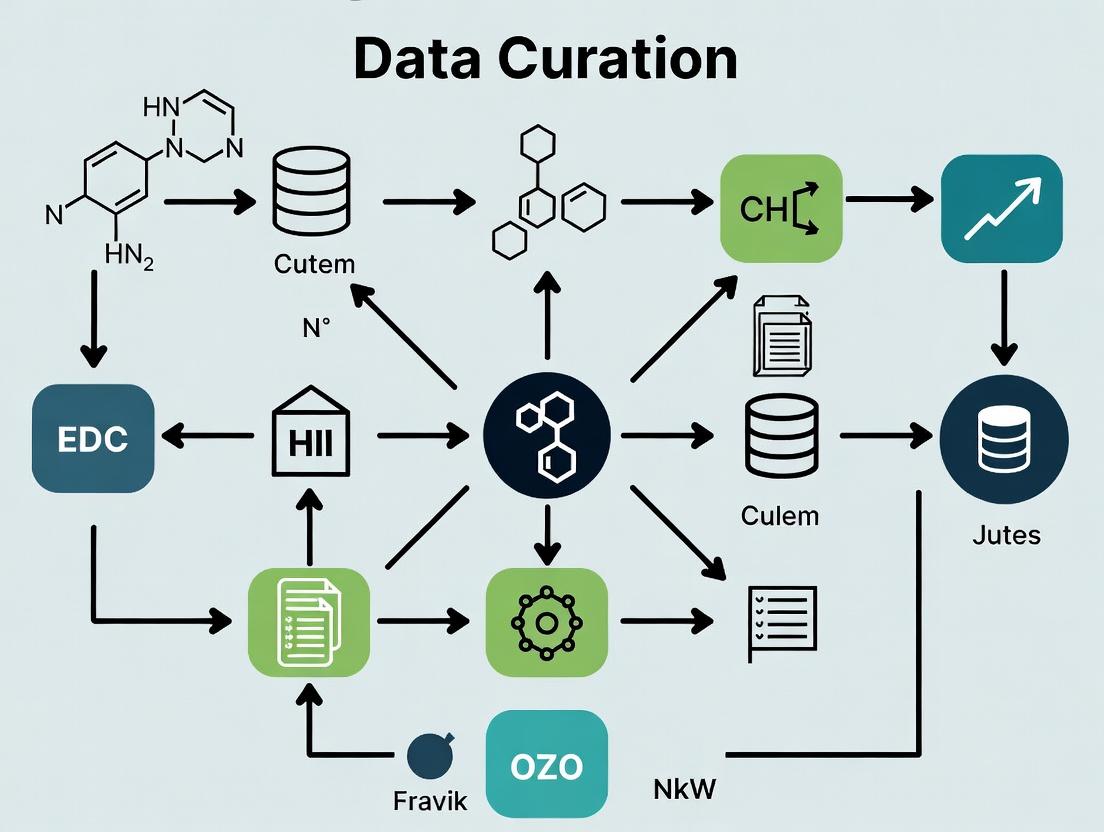
Visualizing the Curation Pipeline and Data Model
The ECOTOX data curation workflow and the logical relationships within its structured database can be visualized as follows.
Diagram 1: ECOTOX Data Curation Pipeline
A systematic, multi-stage process for identifying, reviewing, and ingesting ecotoxicity data.
Diagram 2: ECOTOX Core Data Model
The fundamental entity-relationship structure organizing chemical, species, test, and effect data.
The Scientist's Toolkit: Essential Reagents and Tools for Ecotoxicity Research
While ECOTOX itself is a data resource, the experimental studies it curates rely on a standardized set of materials and tools. The following table details key reagents and solutions fundamental to generating the ecotoxicity data that populates the knowledgebase.
Table 2: Essential Research Reagents & Tools for Ecotoxicity Testing
| Item/Category | Example | Primary Function in Ecotoxicity Studies |
|---|---|---|
| Reference Toxicants | Potassium dichromate, Copper sulfate, Sodium chloride | Used as positive controls to validate test organism health and assay sensitivity. |
| Standardized Test Media | ASTM reconstituted hard water, OECD algal test medium | Provides consistent, defined water chemistry for aquatic tests, ensuring reproducibility. |
| Endpoint Assay Kits | MTT assay (cell viability), ELISA kits (biomarker detection), Chlorophyll a extraction kits | Quantifies specific biological effects, from cytotoxicity in vitro to growth inhibition in algae. |
| Chemical Analysis Standards | Certified reference materials (CRMs) for metals, PAHs, pesticides | Verifies measured exposure concentrations in test solutions, critical for dose-response analysis. |
| Statistical Software | R (with packages like drc for dose-response modeling), USEPA's ToxRStat |
Analyzes toxicity data, calculates EC/LC values, and generates species sensitivity distributions (SSDs). |
The ECOTOX Knowledgebase represents a monumental achievement in environmental data curation. Its value extends far beyond being a simple repository; it is the product of a rigorous, systematic, and transparent process that transforms dispersed scientific literature into a structured, interoperable, and reusable resource. As the demand for rapid chemical safety assessments grows, the role of curated databases like ECOTOX becomes increasingly central. It provides the essential empirical foundation for risk assessment, model development, and the validation of alternative testing strategies, ultimately supporting the protection of ecological health in the face of global chemical challenges.
The continuous introduction of new chemicals into commerce, coupled with expanding regulatory mandates for environmental safety, has created an unprecedented demand for assembled and accessible toxicity data [1]. This need catalyzed the development of the ECOTOXicology Knowledgebase (ECOTOX) by the U.S. Environmental Protection Agency (USEPA) in the early 1980s [1]. Originally conceived as a collection of ecosystem-specific databases for regulatory offices, ECOTOX has evolved into the world’s largest curated compilation of single-chemical ecotoxicity data [1]. Its transformation from a simple archival database to a modern, interactive systematic review platform reflects broader paradigm shifts in toxicology—including the move toward high-throughput in vitro assays, computational modeling, and the adoption of systematic review methods for transparent evidence synthesis [1] [2]. This evolution is central to a thesis on ECOTOX's data curation process, which demonstrates how rigorous, standardized methodologies are critical for generating reliable, reusable data that supports chemical risk assessments, regulatory decisions, and the development of New Approach Methodologies (NAMs) [1] [3].
Historical Development and Architectural Evolution
The development of ECOTOX was driven by practical regulatory needs under statutes like the Clean Water Act and the Toxic Substances Control Act, requiring rapid access to ecological effects data for risk characterization [1]. Its initial architecture in the 1980s consisted of decentralized, taxa-specific databases. The pivotal shift began with the formalization of its data curation pipeline and the adoption of controlled vocabularies, which standardized the extraction of methodological details and results from the literature [1]. The release of ECOTOX Version 5 marks the most significant architectural and philosophical modernization. It introduced a completely redesigned user interface, enhanced query capabilities, and embedded data visualization tools [1] [4]. This version explicitly aligns the database with the FAIR principles (Findable, Accessible, Interoperable, and Reusable), ensuring data can be effectively integrated with other computational toxicology resources and tools [1] [5].
Table 1: The Growth of the ECOTOX Knowledgebase: Key Metrics
| Metric | Historical Scope (1980s-2000s) | Current Scope (ECOTOX Ver 5, 2022-2025) | Data Source |
|---|---|---|---|
| Number of Chemicals | Not specified (focus on pesticides & priority pollutants) | >12,000 chemicals [1] | Peer-reviewed & grey literature |
| Number of Species | Limited, ecosystem-specific | >13,000 aquatic & terrestrial species [4] | Peer-reviewed & grey literature |
| Test Results (Records) | Not specified | >1,000,000 curated test results [1] | >50,000 references [1] |
| Primary Use Case | Internal USEPA regulatory support | Public resource for global research, risk assessment, & model development [1] [4] | N/A |
| Guiding Principles | Data aggregation | FAIR principles & systematic review framework [1] [5] | N/A |
The Systematic Review Framework: Core Methodology
ECOTOX's data curation process is a rigorous, multi-stage pipeline designed to mirror contemporary systematic review practices, ensuring transparency, objectivity, and consistency [1]. The process is governed by detailed Standard Operating Procedures (SOPs) for literature search, citation identification, data abstraction, and data maintenance [1].
The initial phase involves comprehensive searches of both open and "grey" literature (e.g., government reports) for ecologically relevant toxicity studies [1]. Identified references undergo a two-stage screening process: first by title and abstract, followed by a full-text review [1]. For a study to be accepted, it must meet strict applicability and acceptability criteria, which are summarized in Table 2.
Following screening, trained reviewers extract pertinent data from accepted studies using well-established controlled vocabularies. Over 100 data fields are captured, encompassing chemical and species verification, detailed test conditions (exposure duration, concentration, temperature), methodological endpoints, and results [1] [6]. This structured extraction is critical for enabling complex queries and reproducible analyses. The entire workflow, from initial search to data entry, follows a PRISMA-like flow (see Diagram 1), enhancing transparency and minimizing selection bias [1].
Table 2: ECOTOX Study Acceptance Criteria for Data Curation [1] [6]
| Criterion Category | Specific Requirement | Purpose of Criterion |
|---|---|---|
| Test Substance | Single chemical exposure | Ensures clarity of cause-effect relationship |
| Test Organism | Live, whole aquatic or terrestrial plant/animal species | Focus on ecologically relevant endpoints |
| Experimental Design | Reported concurrent environmental concentration/dose & explicit exposure duration | Allows for quantitative dose-response analysis |
| Documented, acceptable control group | Ensures observed effects are treatment-related | |
| Data Reporting | Calculated toxicity endpoint (e.g., LC50, NOEC) is reported or can be derived | Enables data standardization and comparison |
| Study is primary source (not a review) and is publicly available | Ensures data verifiability and traceability | |
| Reporting Standards | Species identified and verified; Test location (lab/field) reported | Assesses relevance and reliability of test conditions |
Diagram 1: ECOTOX Systematic Literature Review and Data Curation Pipeline [1]. The process follows a PRISMA-like flow, with critical screening stages applying the standardized acceptance criteria outlined in Table 2.
Experimental Protocols for Data Generation and Validation
The utility of ECOTOX relies on the quality of the underlying studies from which data is extracted. While ECOTOX itself is a repository, its content generation depends on standardized experimental protocols from primary ecotoxicity research. Key traditional and emerging protocols are highlighted below.
Traditional Whole-Organism Bioassays: The majority of data in ECOTOX comes from standardized in vivo tests, such as the 48-96 hour aquatic acute toxicity test with Daphnia magna (OECD Test Guideline 202) or the fish early-life stage test (OECD TG 210) [2]. These protocols involve exposing organisms to a range of chemical concentrations under controlled conditions to determine lethal or sub-lethal (e.g., growth, reproduction) effects. Key methodological requirements for data inclusion in ECOTOX include specification of exposure medium, temperature, pH, dissolved oxygen, use of appropriate controls, and statistical derivation of endpoints like LC50 (median lethal concentration) [6].
High-Throughput and New Approach Methodologies (NAMs): To address the backlog of untested chemicals, high-throughput screening (HTS) paradigms are emerging [2]. One seminal example is the automated duckweed (Lemna sp.) growth inhibition test. This assay leverages automated image recording and processing to rapidly quantify frond number and area, providing a high-throughput phytotoxicity endpoint [2]. Another advancing area is the use of microfluidic Lab-on-a-Chip (LOC) technologies for small model organisms (e.g., Daphnia, nematodes). These platforms automate animal loading, exposure, and behavioral phenotyping, increasing test throughput while reducing manual labor and animal use [2]. Data from such standardized NAMs, when publicly available, are increasingly curated into repositories like ECOTOX to support model development and validation.
Data Retrieval and Reproducibility Protocols: A critical modern "experimental" protocol is the reproducible retrieval of data from ECOTOX itself. The ECOTOXr R package formalizes this process [5]. The protocol involves: 1) installing the ECOTOXr package in R, 2) using its functions to build targeted API queries (e.g., by chemical CASRN, species name, or effect endpoint), 3) retrieving datasets directly into the R environment, and 4) documenting the entire script for full reproducibility [5]. This tool transforms ad hoc data gathering into a transparent, programmable workflow that aligns with the FAIR principles.
Table 3: The Scientist's Toolkit: Key Reagents and Platforms for Ecotoxicity Research
| Tool/Reagent Category | Specific Example | Primary Function in Ecotoxicity Research | Relevance to ECOTOX & Systematic Review |
|---|---|---|---|
| High-Throughput Bioassay Platforms | Automated imaging systems for Lemna (duckweed) tests [2] | Enables rapid, quantitative assessment of phytotoxicity via frond count and area. | Generates consistent, digital endpoint data suitable for curation and modeling. |
| Microfluidic & Automation Systems | Lab-on-a-Chip (LOC) for Daphnia or nematode bioassays [2] | Automates organism handling, exposure, and real-time behavioral phenotyping. | Increases throughput of in vivo data; provides high-content endpoints for AOP development. |
| Computational Data Access Tools | ECOTOXr R package [5] | Provides programmable, reproducible access to ECOTOX data via API queries. | Embodies FAIR principles; enables transparent and reproducible meta-analysis. |
| Study Evaluation Frameworks | Critical Appraisal Tools (CATs) based on CRED criteria [3] | Provides a structured checklist to assess the reliability and relevance of individual studies. | Supports the systematic review phase of data curation and quality assurance. |
| Reference Chemical Sets | Curated lists of compounds with well-characterized toxicity profiles | Serves as positive controls and benchmarks for calibrating new assay systems. | Provides anchor points for validating NAMs against traditional data within ECOTOX. |
Data Visualization, Interoperability, and the FAIR Framework
ECOTOX Version 5 significantly advanced data accessibility through integrated visualization tools and explicit interoperability features. Users can generate interactive data plots (e.g., scatter plots of effect concentrations) directly within the web interface, allowing for exploratory analysis and identification of trends or outliers [4].
The platform's commitment to the FAIR principles is demonstrated by its interoperability with other major databases. A key integration is with the USEPA's CompTox Chemicals Dashboard, providing seamless linking from a chemical in ECOTOX to rich supplemental data on physicochemical properties, bioactivity, and ongoing toxicological assessments [4]. Furthermore, the development of the ECOTOXr package exemplifies the "Reusable" principle by providing a standardized, script-based method for data retrieval that ensures computational reproducibility [5]. This ecosystem of connected tools (Diagram 2) transforms ECOTOX from a siloed database into a central hub within a broader computational toxicology network, directly supporting the development of Quantitative Structure-Activity Relationship (QSAR) models, species sensitivity distributions (SSDs), and Adverse Outcome Pathways (AOPs) [1] [7].
Diagram 2: ECOTOX Interoperability within the Modern Computational Toxicology Ecosystem. ECOTOX functions as a core data provider, interoperating with chemical (CompTox), mechanistic (AOP), and modeling (QSAR) resources via APIs and linked identifiers. It directly feeds applications in risk assessment and method validation [1] [4] [5].
Future Trajectories: Integrating High-Throughput Data and Advanced Review Methods
The future evolution of ECOTOX will be shaped by two dominant trends in toxicology. First, the expansion of high-throughput and high-content ecotoxicity testing will necessitate the curation of new data types [2]. This includes results from genomic, transcriptomic, and other -omic assays, as well as high-content phenotypic data from automated in vivo platforms [2] [7]. Incorporating such data will require extending controlled vocabularies and developing new modules to capture mechanistic key events, aligning ECOTOX more closely with the Adverse Outcome Pathway (AOP) framework [7].
Second, the systematic review foundation of ECOTOX will be deepened through greater integration of automated screening tools and artificial intelligence. While current screening is manual, future iterations may employ machine learning for title/abstract prioritization and natural language processing to assist in data extraction [2]. Furthermore, the adoption of structured Critical Appraisal Tools (CATs), like those developed by EFSA based on the CRED (Criteria for Reporting and Evaluating Ecotoxicity Data) approach, could be more formally embedded into the curation pipeline to standardize and transparently document reliability and relevance assessments for each study [3].
ECOTOX has completed a transformative evolution from a 1980s internal database to a modern, public systematic review platform. This journey reflects a broader scientific shift towards transparency, reproducibility, and data-driven assessment. Its core strength lies in a rigorous, documented curation process—a systematic review pipeline that applies consistent criteria to identify, evaluate, and extract high-quality ecotoxicity data. By embracing FAIR principles, developing interoperable tools like ECOTOXr, and preparing for next-generation data streams, ECOTOX has established itself as an indispensable infrastructure. It supports not only traditional regulatory risk assessment but also the innovative development and validation of predictive toxicological models and New Approach Methodologies, thereby directly addressing the 21st-century challenge of efficiently evaluating environmental chemical safety.
The systematic assessment of chemical hazards is a cornerstone of environmental protection. For over two decades, the Ecotoxicology (ECOTOX) Knowledgebase has served as a critical infrastructure, transforming dispersed scientific evidence into curated, accessible data to inform regulatory decisions [4]. Its core purpose is to provide a comprehensive, publicly available source of single-chemical toxicity data for ecologically relevant species, thereby supporting the scientific foundation of U.S. environmental statutes [1].
This function is framed within a broader thesis on data curation process research, where ECOTOX exemplifies the application of systematic review principles to ecological toxicology. By implementing a rigorous, transparent pipeline for literature search, study evaluation, and data extraction, ECOTOX ensures that regulatory mandates under laws like the Toxic Substances Control Act (TSCA) and the Clean Water Act (CWA) are met with high-quality, reproducible evidence [1]. For researchers and drug development professionals, understanding this curated data source is vital for designing safer chemicals, evaluating environmental risks of new entities, and developing non-animal New Approach Methodologies (NAMs) that rely on robust historical data for validation [4].
The Regulatory Landscape: TSCA, CWA, and the Need for Curated Data
The regulatory mandates driving chemical assessment are complex and data-intensive. TSCA, as amended by the Lautenberg Act, requires the U.S. Environmental Protection Agency (EPA) to evaluate and manage risks from existing and new chemicals in commerce [8]. Concurrently, the CWA mandates the development of Ambient Water Quality Criteria to protect aquatic life, a process fundamentally reliant on species toxicity data [4]. These laws create a continuous demand for curated, reliable ecotoxicity data.
The ECOTOX Knowledgebase is engineered to meet this demand. It is directly used to inform ecological risk assessments for chemical registration and re-registration, aid in the prioritization and assessment of chemicals under TSCA, and develop numeric criteria for water and sediment quality under the CWA [4]. Recent regulatory proposals, such as the 2025 TSCA Risk Evaluation Framework rule, emphasize efficiency and the use of the best available science, further underscoring the value of centralized, high-quality data repositories like ECOTOX [8].
The ECOTOX Data Curation Pipeline: A Model for Systematic Review
The integrity of ECOTOX is anchored in its meticulous data curation process, which aligns with contemporary systematic review (SR) and evidence-based toxicology practices [1]. This pipeline ensures that the database is not merely a collection of studies but a refined resource of relevant and acceptable toxicity information.
3.1 Experimental Protocol: Literature Search and Study Selection The curation pipeline begins with comprehensive searches of the peer-reviewed and grey literature. Identified references undergo a multi-tiered screening process based on pre-defined applicability and acceptability criteria [1].
Table 1: Key Applicability and Acceptability Criteria for ECOTOX Study Inclusion
| Criterion Category | Description | Example |
|---|---|---|
| Applicability | Relevance to ecological risk assessment. | Test organism is an ecologically relevant aquatic or terrestrial species. |
| Applicability | Study design suitability. | Exposure is to a single, verified chemical stressor. |
| Applicability | Data reporting completeness. | Exposure concentration and duration are explicitly reported. |
| Acceptability | Study reliability and internal validity. | Documented control group is present. |
| Acceptability | Endpoint relevance. | Effect endpoint is clearly defined and measurable (e.g., LC50, NOEC). |
3.2 Data Abstraction and Quality Control Studies passing screening have key details methodically extracted using controlled vocabularies. This includes data on the chemical, test species, exposure conditions, measured effects, and test methodology. Species and chemical identities are verified against authoritative taxonomy and chemistry databases to ensure consistency and interoperability [1]. This rigorous abstraction process transforms narrative journal articles into structured, computable data fields.
3.3 Workflow Visualization The following diagram illustrates the sequential stages of the ECOTOX curation pipeline, from initial search to public data release.
ECOTOX Data Curation and Literature Review Pipeline [1]
Quantitative Scope and Interoperability of the ECOTOX Resource
The scale of curated data within ECOTOX directly reflects its capacity to support broad regulatory and research needs. The knowledgebase is a living resource, updated quarterly with new data [4].
Table 2: Quantitative Summary of the ECOTOX Knowledgebase Scope
| Data Category | Volume | Regulatory and Research Utility |
|---|---|---|
| Scientific References | Over 53,000 compiled references [4]. | Provides an auditable evidence trail for regulatory decisions. |
| Unique Test Records | Over 1,000,000 curated test results [4] [1]. | Enables robust dose-response analysis and meta-analysis. |
| Ecological Species | More than 13,000 aquatic and terrestrial species [4]. | Supports species sensitivity distributions (SSDs) for CWA criteria and ecological risk assessment. |
| Chemical Stressors | Data for over 12,000 chemicals [1]. | Informs assessments across a wide chemical space under TSCA and other statutes. |
ECOTOX enhances its utility through interoperability. It is linked to the EPA CompTox Chemicals Dashboard, which provides additional physicochemical, hazard, and exposure data [4]. This connectivity allows researchers to move seamlessly from a toxicity endpoint in ECOTOX to a chemical's structure, predicted properties, and associated bioassay data, facilitating integrated approaches to safety assessment.
Application in Risk Assessment and New Approach Methodologies (NAMs)
For risk assessors and researchers, ECOTOX data are applied in several critical frameworks. It is fundamental for developing Species Sensitivity Distributions (SSDs), which are used to derive Protective Concentration thresholds for aquatic life [1]. The database also supplies the empirical toxicity data needed to validate and calibrate computational toxicology models, such as Quantitative Structure-Activity Relationship (QSAR) models and ecological thresholds predicted via in vitro to in vivo extrapolation [4].
This role is increasingly important in the context of NAMs. As regulatory science shifts toward reducing vertebrate animal testing, historical in vivo data from ECOTOX becomes essential for anchoring and interpreting high-throughput screening and pathway-based assay results [1]. The database helps identify data gaps, prioritize chemicals for testing, and provide the biological context needed to make mechanistic data ecologically relevant.
The Scientist's Toolkit: Essential Research Reagents and Materials
The experimental studies curated within ECOTOX rely on standardized tools and materials to ensure reproducibility and relevance. The following table details key items central to generating reliable ecotoxicity data.
Table 3: Research Reagent Solutions for Ecotoxicity Testing
| Item | Function in Ecotoxicology | Role in ECOTOX Curation |
|---|---|---|
| Standard Reference Toxicants | (e.g., Sodium chloride, KCl). Used to validate the health and sensitivity of test organisms in a laboratory bioassay. | Studies using reference toxicants for quality control are flagged for higher reliability. |
| Renewal Test Chambers | Flow-through or static-renewal exposure systems for aquatic tests. Control exposure concentration and water quality. | Test system type (static, renewal, flow-through) is a critical extracted field for interpreting exposure dynamics. |
| Formulated Synthetic Water | (e.g., EPA Reconstituted Hard Water). Provides a consistent, defined medium for aquatic toxicity tests, eliminating variability from natural sources. | Water chemistry parameters (hardness, pH, temperature) are extracted as key test condition modifiers. |
| Control Sediments | Defined, uncontaminated sediments for benthic organism testing. Serve as a baseline for assessing toxicity in spiked or field-collected sediments. | The use of appropriate control sediments is a key acceptability criterion for sediment toxicity studies. |
| Standardized Nutrient Media | For algal and aquatic plant toxicity tests (e.g., AAP, OECD media). Ensures consistent growth not limited by nutrients. | Growth medium composition is captured to assess test validity and cross-study comparability. |
Visualizing the Regulatory and Scientific Integration Pathway
The ultimate value of ECOTOX is realized when curated data directly informs regulatory decisions and scientific advancements. The following diagram maps this integration pathway, showing how raw data from controlled studies flow through the knowledgebase to support core regulatory mandates and research initiatives.
Integration of ECOTOX Data into Regulatory and Research Workflows [4] [1] [8]
The ECOTOXicology Knowledgebase (ECOTOX) stands as the world's most comprehensive repository of curated, single-chemical ecotoxicity data [1]. Managed by the U.S. Environmental Protection Agency, this resource is foundational for ecological risk assessment, regulatory decision-making, and environmental research. Its evolution from separate databases in the 1980s to a unified, systematic knowledgebase reflects a commitment to FAIR principles (Findable, Accessible, Interoperable, and Reusable) in toxicological data science [1]. This whitepaper details the technical framework, data curation pipeline, and research applications of ECOTOX, contextualizing its immense scale—over one million test results from more than 12,000 chemicals and 13,000 species—within the rigorous methodology that ensures its reliability and utility for the scientific community [4].
Core Database Metrics and Composition
The ECOTOX Knowledgebase is an ever-expanding resource, updated quarterly with new data extracted from the peer-reviewed and gray literature [4] [9]. Its scale and diversity are summarized in the following tables.
Table 1: Core Quantitative Metrics of the ECOTOX Knowledgebase
| Metric | Count | Description and Source |
|---|---|---|
| Test Records (Results) | >1,000,000 | Individual toxicity test results from acceptable studies [4] [1]. |
| Unique Chemicals | >12,000 | Single, verified chemical stressors, including pesticides, PFAS, and industrial compounds [4] [9]. |
| Ecological Species | >13,000 | Taxonomically verifiable aquatic and terrestrial species [4]. |
| Scientific References | >50,000 | Source publications, including journal articles and technical reports [1]. |
Table 2: Taxonomic Distribution of Test Records (Representative Groups)
| Species Group | Approximate % of Total Records | Key Examples |
|---|---|---|
| Fish | 25.6% | Rainbow trout, Zebrafish, Fathead minnow |
| Flowering Plants/Trees | 18.7% | Duckweed, Soybean, Ryegrass |
| Insects & Spiders | 14.2% | Honey bee, Daphnia magna, Midges |
| Crustaceans | 9.3% | Water flea (Daphnia), Amphipods |
| Mammals | 7.5% | Rat, Mouse, Voles |
| Algae | 5.9% | Green algae, Diatoms |
| Birds | 3.8% | Mallard duck, Bobwhite quail |
| Amphibians | 2.5% | Frog, Toad, Salamander |
Table 3: Diversity of Measured Effects in ECOTOX
| Effect Group | % of Records | Example Endpoints |
|---|---|---|
| Mortality | 26.9% | LC50 (Lethal Concentration to 50%), LD50 |
| Growth | 14.6% | Biomass change, Root elongation inhibition |
| Population | 16.9% | Abundance, Population growth rate |
| Biochemical | 13.8% | Enzyme activity (e.g., AChE inhibition), Hormone levels |
| Physiology | 6.7% | Respiration rate, Photosynthesis efficiency |
| Reproduction | 4.9% | Fecundity, Hatchability, Number of offspring |
| Genetics | 5.2% | Chromosomal aberration, Micronucleus formation |
| Behavior | 3.5% | Avoidance, Feeding rate, Locomotor activity |
| Accumulation | 4.6% | Bioconcentration Factor (BCF), Tissue concentration |
The Data Curation Pipeline: A Systematic Review Protocol
The integrity of ECOTOX is maintained through a formal, multi-stage literature review and data curation pipeline. This process aligns with systematic review methodologies and is governed by detailed Standard Operating Procedures (SOPs) [1].
Experimental Protocol: Literature Search and Screening
The curation workflow is a defined sequence of planning, screening, and extraction.
1. Chemical Identification and Search Strategy: The process begins with the verification of the chemical of interest using its CAS Registry Number (CASRN). Curators compile a comprehensive list of synonyms, trade names, and related chemical forms from sources like the CompTox Chemicals Dashboard and STN [9]. A tailored Boolean search string is constructed and executed across multiple academic databases (e.g., Web of Science, PubMed, Agricola, ProQuest) [10] [1].
2. Screening with PECO Criteria: Identified citations undergo a two-stage screening process against formal PECO (Population, Exposure, Comparator, Outcome) criteria [9].
- Population: Test organisms must be taxonomically verifiable, ecologically relevant species (e.g., fish, plants, invertebrates). Studies on bacteria, viruses, yeast, or human-focused research are excluded [9].
- Exposure: The study must involve exposure to a single, verifiable chemical at quantified concentrations or doses. The route of exposure (e.g., water, diet, injection) and a clear exposure duration must be reported [9].
- Comparator: The study must include an appropriate control treatment for comparison [9].
- Outcome: A measurable biological effect (e.g., mortality, growth reduction) must be reported in relation to the exposure. The source must be a primary research article (not a review) published in English [9].
Studies excluded at the title/abstract or full-text stage are tagged with a specific reason (e.g., "Mixture," "No Concentration," "Review") to ensure transparency and aid process refinement [9].
For studies that pass screening, detailed data are extracted into structured fields using controlled vocabularies to ensure consistency [1].
Key Abstraction Fields:
- Chemical & Species Identifiers: CASRN, DTXSID (EPA's substance identifier), and taxonomic IDs (NCBI Taxonomy, ITIS) [9].
- Test Conditions: Exposure medium, duration, temperature, pH, and test type (acute/chronic).
- Toxicity Results: Endpoint values (e.g., LC50, NOEC, LOEC), effect concentrations, statistical measures, and the author-reported observed duration [10] [9].
- Study Metadata: Reference details, including DOI and ECOTOX-specific reference number (ECOREF).
A single primary study may yield multiple ECOTOX records if it reports results for different species, life stages, or endpoints [9]. All extracted data undergo quality assurance checks before being integrated into the master database and published to the public website in quarterly updates [1] [9].
Integration in Research and Regulatory Applications
Researchers leverage ECOTOX data through a suite of software tools and interoperable resources.
Table 4: Key Research Tools and Resources for ECOTOX Data Analysis
| Tool/Resource Name | Type | Primary Function | Interoperability with ECOTOX |
|---|---|---|---|
| ECOTOXr | R Software Package | Programmatic, reproducible retrieval and curation of ECOTOX data [5]. | Directly queries and processes ECOTOX data exports within the R environment, formalizing the data cleaning pipeline. |
| CompTox Chemicals Dashboard | Interactive Web Application | Provides physicochemical properties, hazard, exposure, and bioactivity data for ~1 million chemicals [11]. | ECOTOX toxicity data is integrated into chemical profiles; linked via DTXSID and CASRN for seamless cross-referencing [11]. |
| USEtox | Scientific Consensus Model | Global model for characterizing human and ecotoxicological impacts in Life Cycle Assessment (LCA) [12]. | ECOTOX data is a critical input for calculating freshwater ecotoxicity characterization factors, particularly for deriving species sensitivity distributions (SSDs) [12]. |
| EPA SSD Toolbox / Web-ICE | Statistical Software Tools | Generate Species Sensitivity Distributions (SSDs) to estimate hazardous concentrations affecting a portion of species [9]. | ECOTOX is a primary data source for constructing SSDs to derive environmental benchmarks like Predicted No-Effect Concentrations (PNECs). |
| R Project & RStudio | Programming Environment | Open-source platform for statistical computing and graphics [10]. | ECOTOX's "Data to R Plot" export function provides customized R scripts and data to regenerate and tailor visualizations from the Explore module [10]. |
Application Workflow: From Data Retrieval to Risk Assessment
A typical research workflow using ECOTOX involves data retrieval, filtration, and synthesis for modeling.
Primary Use Cases:
- Ecological Risk Assessment (ERA): Regulatory bodies use ECOTOX to develop Aquatic Life Criteria, Soil Screening Levels, and Toxicity Reference Values (TRVs) to protect ecosystems [4] [9].
- Chemical Prioritization & Screening: Under laws like the Toxic Substances Control Act (TSCA), data density and potency information from ECOTOX help identify chemicals requiring greater scrutiny [4] [1].
- Model Development and Validation: The curated in vivo data is essential for developing and validating Quantitative Structure-Activity Relationship (QSAR) models, New Approach Methodologies (NAMs), and adverse outcome pathways (AOPs) [4] [1].
- Life Cycle Impact Assessment (LCIA): Models like USEtox rely on ECOTOX data to calculate ecotoxicity characterization factors, translating chemical emissions into potential ecosystem impact scores [12].
Technical Access and Data Export
ECOTOX provides multiple pathways for data access tailored to different user needs [10] [4].
1. Interactive Web Interface:
- Search: For targeted queries with known parameters (specific chemical, species, or endpoint). Users can filter by 19+ parameters, including newly added observed duration filters [10] [4].
- Explore: For open-ended data discovery. Users can browse chemicals, species, or effects and utilize interactive Data Visualization tools with plot exports [10] [4].
- Plot View & R Export: A key feature allows export of plot data paired with an R script to regenerate and customize high-quality graphs externally, facilitating reproducible research [10].
2. Bulk Data Download: The entire database is available for download as pipe-delimited ASCII files, enabling advanced, large-scale analyses [10]. This complete dataset is essential for systematic evidence mapping, large-scale meta-analyses, and integration into other computational platforms.
3. Programmatic Access: The development of the ECOTOXr R package represents a significant advancement toward reproducible and transparent data retrieval, allowing researchers to formally script and document every step of their data curation process [5].
The ECOTOX Knowledgebase is a critical infrastructure component for modern ecotoxicology and environmental chemistry. Its authoritative value stems not merely from its scale—over one million test records for 12,000+ chemicals—but from its rigorous, systematic curation pipeline that adheres to systematic review principles. By implementing FAIR data practices, providing advanced user interfaces, and fostering interoperability with tools like the CompTox Dashboard and USEtox, ECOTOX transforms dispersed literature into actionable, computational-ready knowledge. For researchers and risk assessors, it remains an indispensable resource for deriving protective environmental benchmarks, validating predictive models, and informing the sustainable management of chemicals worldwide. Future developments will continue to enhance its interoperability, computational accessibility, and alignment with evolving paradigms in toxicological assessment.
The ECOTOX Pipeline: A Step-by-Step Guide to Systematic Literature Review and Data Curation
Within the broader research on the ECOTOXicology Knowledgebase (ECOTOX) data curation process, Stage 1: Systematic Literature Searching and Acquisition represents the foundational and critical first phase. ECOTOX is the world’s largest curated database of single-chemical ecotoxicity data, supporting chemical safety assessments and ecological research [1]. Its authority and reliability are directly contingent upon a comprehensive, transparent, and systematic approach to identifying all available evidence. This process is designed to mitigate publication bias—the well-documented tendency for studies showing significant or positive effects to be published more readily than those showing null or negative results [13]. For a definitive resource like ECOTOX, which informs regulatory decisions under statutes like the Clean Water Act and the Toxic Substances Control Act [4], failing to capture this "grey literature" would result in a skewed, non-representative dataset. This guide details the technical methodology of this initial stage, framing it as an essential component of a robust, evidence-based data curation pipeline that ensures the ECOTOX knowledgebase remains a FAIR (Findable, Accessible, Interoperable, and Reusable) resource for the global scientific and regulatory community [1].
Defining the Search Universe: Open and Grey Literature
A systematic search for ECOTOX data curation explicitly targets two broad domains: traditional open literature and grey literature.
Open Literature: This refers to commercially published, peer-reviewed scientific material typically indexed in major bibliographic databases (e.g., PubMed, Scopus, Web of Science). It includes journal articles, published reviews, and academic monographs.
Grey Literature: Defined as literature produced by entities outside of traditional commercial or academic publishing channels [14]. For ecotoxicology, this encompasses:
- Government and Agency Reports: Technical reports from environmental protection agencies (e.g., U.S. EPA, Environment Canada), health departments, and international bodies like the World Health Organization (WHO) [14].
- Academic Work: Doctoral dissertations and master's theses, which often contain extensive original data [14].
- Conference Proceedings: Abstracts, posters, and full papers presented at scientific conferences [13].
- Regulatory and Trial Data: Unpublished or ongoing study reports from chemical manufacturers, and records from clinical and ecological trial registries [14] [13].
- Preprints: Preliminary versions of research articles shared on servers like bioRxiv and arXiv prior to peer review [14].
The inclusion of grey literature is not optional; it is a scientific imperative. Studies suggest that papers with "interesting" results are three times more likely to be published [13]. Relying solely on open literature risks creating a "file-drawer" problem, where an incomplete and positively biased evidence base leads to inaccurate hazard assessments [13]. A classic example is the antidepressant Agomelatine, where a review of both published and unpublished trials revealed a more modest efficacy profile and underreported safety concerns than the published literature alone suggested [13].
Quantitative Scope of ECOTOX Data Curation
The scale of the ECOTOX knowledgebase underscores the importance of a rigorous Stage 1 search protocol. The following table summarizes the current quantitative scope of the curated data, which is the direct product of systematic literature searching and acquisition [4] [1].
Table 1: Quantitative Scope of the ECOTOX Knowledgebase (as of 2025)
| Data Category | Metric | Description |
|---|---|---|
| Total References | Over 53,000 | The number of individual source documents (from both open and grey literature) from which data has been curated [4] [1]. |
| Curated Test Records | Over 1,000,000 | Individual toxicity test results extracted and entered into the knowledgebase [4]. |
| Chemical Coverage | Over 12,000 | Unique single chemical stressors with associated toxicity data [4] [1]. |
| Species Coverage | Over 13,000 | Ecologically relevant aquatic and terrestrial species represented in the database [4]. |
Experimental Protocol: The ECOTOX Literature Review Pipeline
The ECOTOX team employs a documented, multi-stage pipeline for literature review and data curation that aligns with systematic review principles [1]. The workflow for Stage 1 and initial screening is visualized below.
Diagram 1: ECOTOX Literature Search and Screening Workflow (Max Width: 760px)
Detailed Methodological Steps
Strategy Development (Protocol): For each chemical or project, a structured search protocol is defined. This includes:
- Population/Test System: Ecologically relevant species (aquatic and terrestrial).
- Stressor: Single, verified chemical substances.
- Outcome: Measured toxicity endpoints (e.g., mortality, growth, reproduction).
- Search Strings: Boolean logic-based queries incorporating chemical names, synonyms, CAS numbers, and broad toxicity terms, tailored for each database [1].
Search Execution: Searches are performed across multiple sources concurrently [1].
- Open Literature Databases: PubMed/MEDLINE, Scopus, Web of Science, Environmental Sciences and Pollution Management.
- Grey Literature Sources: As detailed in Section 5 (The Scientist's Toolkit). This includes targeted searches in government repositories, thesis databases, and trial registries.
Title/Abstract Screening: Retrieved references are independently screened by two reviewers against pre-defined applicability criteria. These criteria determine if a study is within scope (e.g., original ecotoxicity data, relevant species and chemical, controlled experiment) [1]. Conflicts are resolved by consensus or a third reviewer.
Full-Text Review and Acceptability Screening: The full text of potentially applicable studies is obtained and assessed against more detailed acceptability criteria. This quality assessment evaluates study reliability, focusing on factors like documented methodology, appropriate controls, and clear reporting of results and raw data [1]. Studies failing to meet minimum quality thresholds are excluded.
Data Extraction Ready Set: The final output of Stage 1 is a vetted set of high-quality, relevant studies that proceed to the next stage: structured data abstraction into the ECOTOX knowledgebase.
Success in grey literature search requires knowing where to look. The following table catalogs essential resources and their function within the ecotoxicology data curation context [14] [13].
Table 2: Research Reagent Solutions for Grey Literature Acquisition
| Resource Category | Resource Name | Function in ECOTOX Data Curation |
|---|---|---|
| Theses & Dissertations | ProQuest Dissertations & Theses Global [14] | Locates foundational academic research containing extensive raw data not always published elsewhere. |
| EThOS (British Library) [14] | Provides access to UK doctoral theses. (Note: Temporarily offline as of 2023) [14]. | |
| Open Access Theses and Dissertations (OATD) [13] | Searches globally for freely available graduate theses. | |
| Government & Agency Repositories | WHO IRIS (Institutional Repository) [14] | Sources international technical reports and policy documents on chemical safety and health. |
| U.S. EPA Web Portal [4] | Primary source for EPA technical reports, risk assessments, and data relevant to U.S. regulations. | |
| World Bank Open Knowledge Repository [14] | Provides reports on environmental projects and chemical impacts in developing regions. | |
| Clinical & Ecological Trial Registries | ClinicalTrials.gov [14] | Identifies unpublished, ongoing, or completed studies on chemical effects, including non-human subjects. |
| WHO ICTRP (Intl. Clinical Trials Registry) [14] | A global portal searching across national trial registries. | |
| EU Clinical Trials Register [14] | Source for trial information within the European Union. | |
| Preprint Servers | bioRxiv [14] | Discovers cutting-edge, non-peer-reviewed research in biology and toxicology. |
| arXiv [14] | Covers quantitative biology, physics, and related computational fields relevant to model development. | |
| Specialized Grey Lit Databases | Grey Matters (CADTH) [14] | A practical checklist and tool for identifying health-related grey literature sources. |
| Global Index Medicus (WHO) [14] | Focuses on biomedical literature from low- and middle-income countries. |
Data Flow and Interoperability Post-Acquisition
The conclusion of Stage 1 initiates the critical data curation and integration phases. The relationships between acquired data, the ECOTOX knowledgebase, and downstream applications are complex and bidirectional, supporting both regulatory assessment and predictive modeling.
Diagram 2: Data Curation Flow and Interoperability from Search to Application (Max Width: 760px)
As shown, the acquired and curated data serves multiple high-value purposes:
- Direct Query and Visualization: Through the ECOTOX interface, users can search, filter, and visualize data via interactive plots [4].
- Support for New Approach Methodologies (NAMs): Curated in vivo data is essential for developing and validating computational toxicology models, such as Quantitative Structure-Activity Relationships (QSARs) and Species Sensitivity Distributions (SSDs) [4] [1].
- Informing Regulatory Standards: Data directly feeds into the derivation of chemical benchmarks, water quality criteria, and ecological risk assessments [4].
- Closes the Research Loop: The use of ECOTOX in modeling and assessment continuously identifies data gaps for specific chemicals or species, which in turn informs and prioritizes future systematic search efforts (Stage 1), creating an iterative, evidence-driven cycle of knowledge refinement [1].
Stage 1: Systematic Literature Searching and Acquisition is a meticulously engineered process that underpins the scientific integrity of the ECOTOX knowledgebase. By employing a protocol-driven, dual-path approach that rigorously targets both open and grey literature, the ECOTOX curation pipeline actively combats publication bias and strives for comprehensiveness. This results in a foundational dataset that is not only massive in scale—exceeding one million test records—but also balanced and reliable [4] [1]. As outlined, this stage is the critical first link in a chain that transforms disparate research findings into a structured, interoperable, and FAIR resource. This resource, in turn, accelerates ecological risk assessment, fuels the development of predictive toxicological models, and ultimately supports informed decision-making to protect environmental and public health.
Within the broader thesis on the ECOTOXicology Knowledgebase (ECOTOX) data curation process research, Stage 2 represents the critical juncture where identified scientific literature is systematically evaluated for inclusion. This stage transforms a collection of potential references into a curated body of evidence suitable for ecological risk assessment and regulatory decision-making. ECOTOX, as the world's largest compilation of curated ecotoxicity data, relies on a transparent and repeatable screening protocol to ensure data quality, consistency, and relevance for its over one million test results from more than 50,000 references [1]. This guide details the technical execution of Stage 2, providing researchers, scientists, and drug development professionals with an in-depth analysis of the defined applicability and acceptability criteria, experimental protocols, and quality control measures that underpin this authoritative resource.
Defining Applicability and Acceptability Criteria
The screening process bifurcates into two sequential assessments: applicability (relevance) and acceptability (quality). These criteria are derived from standardized evaluation guidelines and are fundamental to systematic review practices [1] [6].
Core Applicability Criteria
Applicability determines if a study investigates a question relevant to the knowledgebase's scope. A study must meet all of the following minimum criteria to be considered applicable [1] [6]:
- Single Chemical Exposure: Effects must be attributable to exposure to a single, identifiable chemical.
- Ecologically Relevant Species: Test subjects must be whole, live aquatic or terrestrial plants or animals.
- Measured Biological Effect: The study must report a quantifiable biological effect on the organism.
- Reported Exposure Concentration/Dose: A concurrent environmental chemical concentration, dose, or application rate must be specified.
- Explicit Exposure Duration: The duration of chemical exposure must be clearly stated.
Core Acceptability Criteria
Acceptability assesses the methodological soundness and reporting quality of an applicable study. These criteria ensure data verifiability and robustness [6].
Table 1: Quantitative Summary of ECOTOX Knowledgebase (as of 2022 Publication)
| Metric | Count | Description |
|---|---|---|
| Curated Chemicals | >12,000 | Unique chemical substances with ecotoxicity data. |
| Ecological Species | >N/A (implied thousands) | Aquatic and terrestrial species represented. |
| Test Results | >1,000,000 | Individual toxicity endpoint records. |
| Source References | >50,000 | Scientific papers, reports, and studies curated. |
The Stage 2 Screening Workflow: A Systematic Protocol
The screening process follows a defined pipeline with sequential gates, ensuring efficiency and consistency. The workflow is visually summarized in Figure 1.
Diagram 1: ECOTOX Literature Screening and Data Curation Workflow
Experimental Protocol for Screening
The protocol is executed by trained reviewers following detailed Standard Operating Procedures (SOPs) [1].
Title and Abstract Screen:
- Objective: To rapidly exclude clearly irrelevant literature.
- Method: Reviewers assess the title and abstract against the five core applicability criteria. Studies not involving ecologically relevant species, single-chemical toxicity, or reporting of an effect and exposure are rejected.
Full-Text Acquisition and Initial Review:
- Objective: To obtain the complete study for definitive evaluation.
- Method: The full text of all potentially relevant studies is retrieved. An initial review confirms basic relevance and checks for major exclusion factors (e.g., non-English language, not a full primary article) [6].
Formal Applicability Assessment:
- Objective: To definitively judge if the study falls within ECOTOX's scope.
- Method: Using a standardized checklist, reviewers verify the presence of all five core applicability elements within the full text. Studies failing any criterion are documented and excluded.
Formal Acceptability Assessment:
- Objective: To evaluate the methodological quality and reporting completeness of applicable studies.
- Method: Reviewers apply the acceptability criteria, focusing on:
- Control Groups: Verification of concurrent, untreated or vehicle control groups for comparison [6].
- Endpoint Calculation: Confirmation that a quantitative toxicity endpoint (e.g., LC50, NOEC) is reported or can be calculated from provided data [6].
- Reporting Standards: Assessment of whether key test conditions (species identification, temperature, pH, test location) are unambiguously reported.
Documentation and Resolution:
- All decisions are recorded in a tracking system. Uncertain cases are escalated for consensus review by senior curators to maintain consistency.
The Scientist's Toolkit: Research Reagent Solutions
The following tools and resources are essential for executing or understanding the Stage 2 screening process.
Table 2: Essential Toolkit for ECOTOX Data Screening and Curation
| Item | Function in Screening Process | Relevance for Researchers |
|---|---|---|
| Controlled Vocabularies & Taxonomies | Standardize terminology for species, chemicals, and endpoints during data extraction, ensuring interoperability and searchability [1]. | Critical for aligning in-house data with ECOTOX structure for comparison or submission. |
| Chemical Verification Tools (e.g., CAS RN, InChIKey) | Unambiguously identify the tested chemical, separating it from metabolites or mixtures, a key applicability criterion [1]. | Prevents misidentification in literature reviews; essential for QSAR and computational modeling. |
| Species Verification Databases | Confirm the taxonomic identity and ecological relevance of test organisms [1]. | Ensures accurate extrapolation of toxicity data across related species in risk assessment. |
| Systematic Review Software (e.g., for PRISMA) | Manage the flow of references, track screening decisions, and generate audit trails, as reflected in ECOTOX's internal SOPs [1]. | Provides transparency and reproducibility for independent systematic reviews in ecotoxicology. |
| EPA ECOTOX Knowledgebase (Public Interface) | Serves as the public portal for accessing the final curated data output of the screening process [1]. | Primary source for retrieving quality-controlled ecotoxicity data for chemical assessments and research. |
Integration with Broader Assessment Frameworks
The output of Stage 2 screening directly feeds into higher-order ecological risk assessments. A primary application is in developing Species Sensitivity Distributions (SSDs), which are used to derive protective benchmarks like Ecological Soil Screening Levels (Eco-SSLs) [1] [15].
Diagram 2: Pathway from Literature Screening to Protective Benchmarks
The rigorous screening in Stage 2 ensures that only relevant and reliable data populate the SSD, leading to scientifically defensible environmental safety values. This underscores the critical role of structured curation in supporting regulatory science and the assessment of chemical safety under mandates like the Endangered Species Act [6].
Within the context of a broader thesis on the ECOTOX knowledgebase data curation process research, Stage 3 represents the critical implementation phase where systematic review principles are operationalized. The Ecotoxicology (ECOTOX) Knowledgebase, maintained by the United States Environmental Protection Agency (USEPA), is the world's largest curated repository of single-chemical toxicity data for ecological species [1]. Its utility in regulatory risk assessments, chemical prioritization under statutes like the Toxic Substances Control Act (TSCA), and ecological research hinges on the consistency, reliability, and findability of its over one million test records [4].
This stage transforms the screened and accepted scientific evidence, identified through exhaustive literature searches, into structured, computable data. Data abstraction is the meticulous process of extracting pertinent information from source studies, while controlled vocabulary is the standardized language that ensures uniform entry and retrieval. Together, they form the backbone of a FAIR (Findable, Accessible, Interoperable, and Reusable) data resource, enabling sophisticated queries, interoperability with tools like the CompTox Chemicals Dashboard, and support for quantitative modeling such as species sensitivity distributions (SSDs) and quantitative structure-activity relationship (QSAR) models [4] [1]. This guide details the technical protocols and systems that underpin this transformation, providing a framework for high-fidelity data curation in ecotoxicology.
Data abstraction is the targeted extraction of specific data points and metadata from a primary research article into a structured field-based format. In ECOTOX, this moves beyond simple digitization to capture the nuanced context of each toxicity test. The process is governed by detailed Standard Operating Procedures (SOPs) designed to minimize curator subjectivity and maximize consistency [1]. Key abstracted elements include:
- Chemical Identity: Verified substance name, CAS Registry Number, and molecular details.
- Species Taxonomy: Verified organism identity, including Latin binomial, life stage, and source.
- Experimental Design: Exposure pathway, duration, concentration/dose levels, media, and temperature.
- Control Conditions: Description and performance of control groups.
- Endpoint Results: Quantitative toxicity measures (e.g., LC50, EC10, NOEC) with reported values, statistical significance, and units.
Controlled Vocabulary: The Architecture of Consistency
A controlled vocabulary is a prescriptive, organized set of terms and phrases used for indexing and retrieval, where one preferred term is designated for each concept [16]. Its primary function is vocabulary control, which suppresses the "anarchy of natural language" by managing synonyms, distinguishing homographs, and identifying semantic relationships between terms (e.g., broader, narrower, related) [16] [17].
Types of Controlled Vocabularies Relevant to ECOTOX:
- Simple Term Lists (Pick Lists): Used for fields with a limited set of unambiguous options (e.g., exposure media: "Freshwater," "Saltwater," "Sediment") [16].
- Thesauri: Provide semantic relationships and are ideal for complex concepts like toxicological effects (e.g., linking "Mortality" to related terms like "Lethality") [17].
- Taxonomies & Ontologies: Hierarchical systems that may be used for structuring species kingdoms or chemical classes, supporting more advanced computational reasoning.
The implementation of a controlled vocabulary ensures that all curators describe the same experimental condition using the same term (e.g., "Salmo salar" instead of "Atlantic salmon," "young adult," or "smolt"), enabling precise data collocation and retrieval [16].
The ECOTOX Data Curation Pipeline: A Stage 3 Protocol
The following workflow details the sequential steps for abstracting data and applying controlled vocabulary, from the receipt of an accepted study to its entry into the knowledgebase.
Diagram: Sequential Workflow for Data Abstraction and Vocabulary Curation. This protocol ensures systematic processing from an accepted study to a validated database record.
Step 1: Full-Text Review & Critical Appraisal The curator performs a detailed read of the complete study to understand the experimental narrative fully. This step verifies that the study meets all Phase II acceptability criteria, including the use of an appropriate control, clear reporting of results, and a defensible endpoint calculation [6]. Studies are classified for their potential use in risk assessment (e.g., definitive screening, limit test).
Step 2: Chemical Verification & Standardization The chemical stressor is identified and linked to a verified, unique identifier. This process typically involves:
- Matching the reported chemical name to a master chemical list (e.g., via the EPA CompTox Chemicals Dashboard).
- Resolving ambiguities (e.g., "BPA" to "Bisphenol A").
- Recording the definitive CASRN (Chemical Abstracts Service Registry Number) and structure information. This creates interoperability with other chemical databases [1].
Step 3: Species Verification & Taxonomic Alignment The test organism is verified using authoritative taxonomic databases (e.g., Integrated Taxonomic Information System - ITIS). The Latin binomial (genus, species) is standardized, and relevant life stage, age, or sex data is captured. This ensures data for "Oncorhynchus mykiss" is distinct from "Danio rerio," regardless of the common names used in the source paper.
Step 4: Experimental Data Abstraction Quantitative and qualitative data are extracted into predefined fields. This includes [1]:
- Test Conditions: Exposure duration, concentrations/doses, media chemistry (pH, hardness), temperature.
- Endpoint & Statistical Results: The specific toxicity metric (e.g., LC50 value, its confidence intervals, the statistical test used).
- Effect Measurement: The observed biological effect (e.g., mortality, growth inhibition, reproduction impairment) linked to the endpoint.
Step 5: Application of Controlled Vocabulary The curator translates the author's narrative into the knowledgebase's standardized language using pick lists, thesauri, and authority files [16]. For example:
- An author's description of "eggs didn't hatch" is coded as the effect "Hatching Success."
- A test described as a "96-hour flow-through acute test" is tagged with the terms "Acute," "Flow-Through," and the duration "96 h."
- The species life stage "juvenile" is mapped to a precise term like "Juvenile (specified)."
Step 6: Quality Assurance & Validation Check The abstracted record undergoes automated and/or peer validation. Automated checks may flag outliers or missing required fields. A second curator may review a subset of records to ensure adherence to SOPs and consistency in vocabulary application. Failed records are flagged and returned to Step 1 for correction [1].
Core Controlled Vocabulary Structures in ECOTOX
The knowledgebase employs a multi-layered vocabulary system to describe the core entities and concepts.
Chemical Hierarchy & Identity
Chemicals are organized by identity, not function. The primary vocabulary includes preferred chemical names, synonyms, and unique identifiers (CASRN, DTXSID from CompTox). A chemical ontology may classify them by structure (e.g., "Polycyclic Aromatic Hydrocarbons") to support QSAR modeling [4].
Diagram: Hierarchical Vocabulary Structure for Chemical Identity Standardization.
Taxonomic & Biological Effect Vocabularies
- Species Vocabulary: A taxonomy-driven hierarchy (Kingdom > Phylum > Class > Order > Family > Genus > Species) ensures precise organism identification. Common names are included as non-preferred terms pointing to the Latin binomial [16].
- Effect/Endpoint Vocabulary: A thesaurus organizes biological responses. Broader terms (e.g., "Reproduction") have narrower terms (e.g., "Fecundity," "Hatching Success"). Related terms (e.g., "Growth" and "Development") are cross-linked to aid discovery [17].
Quantitative Framework: Acceptance Criteria and Data Scale
The curation process is governed by explicit, binary criteria that determine a study's acceptability for abstraction.
Table 1: Phase I Minimum Acceptance Criteria for Data Abstraction [6]
| Criterion Number | ECOTOX Acceptance Requirement | Rationale for Curation |
|---|---|---|
| 1 | Single chemical exposure | Maintains focus on causative agent for use in chemical-specific assessments. |
| 2 | Effect on aquatic/terrestrial plant or animal | Ensures ecological relevance. |
| 3 | Biological effect on live, whole organism | Excludes in vitro cellular studies (though relevant for NAMs context). |
| 4 | Concurrent chemical concentration/dose reported | Essential for dose-response modeling and benchmark derivation. |
| 5 | Explicit exposure duration reported | Critical for distinguishing acute from chronic effects. |
| 6 | Chemical of concern to OPP (for regulatory assessments) | Ensures regulatory utility. |
| 7 | Article published in English | Practical limitation for curation. |
| 8 | Study presented as a full article | Ensures sufficient methodological detail is available. |
| 9 | Publicly available document | Promotes transparency and verifiability. |
| 10 | Paper is the primary source of data | Avoids duplication and potential transcription errors. |
| 11 | A calculated endpoint is reported (e.g., LC50, NOEC) | Provides a standardized, quantitative metric for comparison. |
| 12 | Treatment(s) compared to an acceptable control | Establishes a baseline for determining treatment-related effects. |
| 13 | Study location (lab/field) reported | Provides context for interpreting environmental relevance. |
| 14 | Tested species is reported and verified | Fundamental for species-specific analysis and SSD development. |
Table 2: Scale of Curated Data in ECOTOX Knowledgebase (as of 2025) [4] [1]
| Data Category | Quantitative Scale | Significance for Research |
|---|---|---|
| Total References | > 53,000 | Represents the comprehensive scope of the systematically searched literature. |
| Total Test Results | > 1,000,000 | Indicates the granularity of data available for meta-analysis and modeling. |
| Unique Chemicals | ~12,000 - 13,000 | Demonstrates broad chemical coverage for comparative hazard assessment. |
| Ecological Species | > 13,000 (aquatic & terrestrial) | Enables the development of robust Species Sensitivity Distributions (SSDs). |
| Data Update Frequency | Quarterly | Ensures the knowledgebase remains current (an "evergreen" resource). |
Table 3: Key Research Reagent Solutions and Curation Tools
| Category | Tool / Resource | Primary Function in Curation/Research |
|---|---|---|
| Chemical Identity | EPA CompTox Chemicals Dashboard | Authoritative source for chemical verification, CASRN/DTXSID mapping, and obtaining related physicochemical properties [4] [1]. |
| Taxonomic Verification | Integrated Taxonomic Information System (ITIS) | Standard reference for validating species nomenclature and taxonomic hierarchy [1]. |
| Bibliographic Management | Reference Databases (e.g., PubMed, Web of Science) | Sources for conducting systematic literature searches using controlled vocabulary (e.g., MeSH terms) and Boolean operators [17]. |
| Controlled Vocabulary | Custom ECOTOX Thesauri & Pick Lists | Internal standardized lists for effects, endpoints, test conditions, and other critical fields to ensure curator consistency [1]. |
| Data Quality & Modeling | Quantitative Structure-Activity Relationship (QSAR) Software | Uses curated toxicity data from ECOTOX to develop and validate predictive models for chemical prioritization [4]. |
| Statistical Analysis | Species Sensitivity Distribution (SSD) Generators (e.g., ETX 2.0, SSD Master) | Analyzes curated toxicity data across multiple species to derive protective environmental thresholds (e.g., HC5) [1]. |
| Accessibility & Compliance | Color Contrast Checkers (e.g., based on WCAG 2.2 guidelines) | Ensures that any visualizations or interfaces developed for data presentation meet enhanced contrast requirements (≥7:1 for standard text) for accessibility [18] [19]. |
Stage 3 of the ECOTOX curation pipeline—data abstraction paired with rigorous controlled vocabulary application—transforms peer-reviewed literature into a robust, computable knowledge asset. This process, framed within systematic review practices, directly supports the evolving paradigm in toxicology. The resulting high-quality, standardized data is indispensable for validating New Approach Methodologies (NAMs), training machine learning models, and conducting transparent chemical safety assessments. By adhering to the technical protocols and principles outlined in this guide, the knowledgebase not only preserves the value of legacy animal testing but also provides the essential empirical foundation required to advance predictive ecotoxicology in the 21st century.
The data curation process for the ECOTOX Knowledgebase is a systematic, multi-stage operation designed to transform raw ecotoxicological literature into a FAIR (Findable, Accessible, Interoperable, and Reusable) scientific resource. This process is the core subject of a broader research thesis on scalable environmental data management [20]. Stage 4, encompassing Data Maintenance, Quarterly Updates, and Public Release, represents the final, continuous cycle of this pipeline. It is where curated data achieves operational utility and public accessibility. In ECOTOX Version 5, this stage has been significantly enhanced to support the needs of modern chemical risk assessment and research, which demand timely, transparent, and interoperable data [20].
The primary functions of Stage 4 are threefold: to maintain the integrity and accuracy of over one million existing test records; to integrate newly curated data from the ongoing literature review process on a quarterly schedule; and to publicly release this data through a redesigned web interface and API, ensuring it is actionable for regulatory decision-makers, researchers, and model developers [4] [20].
Core Components of Data Maintenance
Data maintenance is the foundational activity that ensures the long-term reliability and consistency of the knowledgebase. It involves systematic processes to preserve data quality and adapt to evolving scientific standards.
Back-End Data Management and Version Control: The core ECOTOX data is maintained in a relational database structure, where tables for chemicals, species, tests, and results are linked via unique identifiers [21]. A rigorous version control system tracks all changes to the underlying data. This is critical for reproducibility, allowing users to reference specific data releases (e.g., the September 2022 release used to build the ADORE machine learning benchmark dataset) [21]. Archived versions of related databases, such as ToxValDB, are also maintained to provide a historical record [22].
Vocabulary and Standardization Maintenance: Consistency is enforced through the use of controlled vocabularies for key fields such as chemical names, species taxonomy, test media, and measured effects [20] [6]. Maintenance involves curating these vocabularies, adding new terms as needed by emerging science, and mapping legacy terms to current standards. This standardization is what enables precise searching and large-scale data aggregation.
Linkage and Interoperability Updates: A key maintenance task is updating and validating links to external resources. Each chemical is associated with identifiers like the DSSTox Substance ID (DTXSID), which links directly to the EPA's CompTox Chemicals Dashboard for rich chemical property data [4] [21]. Maintaining these linkages ensures ECOTOX remains an interoperable node within a larger network of computational toxicology resources [22] [23].
Protocol for Quarterly Update Cycles
The quarterly update is a scheduled, structured process for expanding the knowledgebase with newly curated information. The protocol ensures that each update is consistent, traceable, and seamlessly integrated.
1. Literature Acquisition and Curation Window: Prior to each quarterly release, a defined period (e.g., the previous six months) is established for processing newly published literature. The ECOTOX team performs comprehensive searches of scientific databases, identifies relevant studies on single-chemical toxicity to ecological species, and applies established systematic review procedures for study evaluation and data extraction [20].
2. Data Validation and Integration Batch Processing: Extracted data from newly accepted studies undergoes a multi-tier validation check. This includes automated checks for format and required fields, as well as expert manual review for scientific accuracy and proper application of controlled vocabularies. Validated data is then formatted into standard batches for integration into the main database tables [6] [21].
3. Pre-Release Quality Assurance (QA): Before public deployment, the updated database undergoes a comprehensive QA process. This involves running automated test queries to verify data integrity, checking a sample of new entries for accuracy, and ensuring that all search, filtering, and visualization functions perform correctly with the new data. The system's interoperability with linked tools like the CompTox Chemicals Dashboard is also verified [4].
4. Version Documentation and Release Notes: Each quarterly update is assigned a discrete version identifier. Detailed release notes are generated, documenting the number of new references, tests, and chemicals added, as well as any changes to the user interface, underlying vocabularies, or API functionality [20]. This mirrors the transparent update practices of other EPA data tools [24].
The following diagram illustrates this cyclical workflow:
Diagram 1: ECOTOX Quarterly Data Update Workflow - This flowchart depicts the staged, iterative process for integrating new curated data into the public knowledgebase.
Table 1: Metrics of the ECOTOX Knowledgebase (as of 2022 Release)
| Data Category | Count | Description and Source |
|---|---|---|
| Total References | >53,000 | Peer-reviewed sources from systematic literature searches [4] [20]. |
| Total Test Results | >1,000,000 | Individual toxicity records extracted from accepted studies [4] [20]. |
| Unique Chemicals | ~12,000 | Single chemical stressors, linked to DSSTox IDs [20] [21]. |
| Unique Species | >13,000 | Aquatic and terrestrial plant and animal species [4]. |
| Data Update Frequency | Quarterly | Scheduled release of newly curated data [20]. |
Table 2: Example Quarterly Update Metrics (Illustrative Scope)
| Update Component | Typical Volume per Quarter | Maintenance Action |
|---|---|---|
| New References Added | Hundreds | Integrated into searchable bibliography. |
| New Test Results Added | Thousands | Added to main data tables with unique result IDs [21]. |
| New Chemical-Species Pairs | Variable | New relationships established in database. |
| Vocabulary Updates | As needed | Controlled lists for effects, media, etc., are expanded. |
Public Release Architecture in Version 5
ECOTOX Version 5 introduced a completely redesigned public interface and backend architecture focused on user accessibility, data exploration, and interoperability [20].
Redesigned User Interface (UI) and Enhanced Query Functions: The public web interface offers three primary access modes [4]:
- Search: For targeted queries using specific chemical, species, or effect parameters with 19 refineable filters.
- Explore: For open-ended discovery when exact parameters are unknown.
- Data Visualization: Interactive plotting tools that allow users to visualize dose-response trends and explore data relationships graphically.
Application Programming Interface (API) for Programmatic Access: To support advanced research and integration into automated workflows, ECOTOX data is accessible via the EPA's Computational Toxicology and Exposure (CTX) APIs [23]. These "open data" APIs allow users to programmatically retrieve specific data subsets, enabling direct integration with computational modeling pipelines and custom applications. Access requires a free API key [23].
Customizable Data Export and Interoperability: Users can customize output fields (from over 100 available) for export in machine-readable formats. The system's interoperability is demonstrated by its direct linkage to the CompTox Chemicals Dashboard for chemical information and by its use as the primary source for curated data in downstream resources, such as the ADORE benchmark dataset for machine learning in ecotoxicology [4] [21].
Quality Assurance and Validation Protocols
Quality assurance is embedded throughout Stage 4, governed by formal guidelines to ensure data is fit for regulatory and research purposes [6].
Adherence to EPA Evaluation Guidelines: The Evaluation Guidelines for Ecological Toxicity Data in the Open Literature provide the definitive protocol for OPP risk assessors using ECOTOX data [6]. These guidelines formalize the acceptance criteria for studies, requiring, for example, reported exposure durations, concurrent controls, and verified species identification. Stage 4 maintenance ensures the live database reflects these standards.
Systematic Review Alignment: The curation process is designed to align with systematic review practices, incorporating transparent literature search, objective study evaluation, and consistent data extraction [20]. This methodological rigor is maintained during quarterly updates to ensure new data meets the same evidence-based standard.
Validation Through Downstream Application: The ultimate validation of ECOTOX data quality is its successful application in high-stakes contexts. It is the primary source for developing national water quality criteria, informing Endangered Species Act assessments, and serving as the empirical foundation for Quantitative Structure-Activity Relationship (QSAR) and machine learning models aimed at reducing animal testing [4] [20] [21]. The ongoing global revision of statistical standards, such as the OECD No. 54 document on ecotoxicity data analysis, also informs best practices for deriving and using the endpoints stored in ECOTOX [25].
Table 3: Key Research Reagent Solutions & Tools for ECOTOX Data Utilization
| Tool/Resource Name | Primary Function | Relevance to ECOTOX Research |
|---|---|---|
| CompTox Chemicals Dashboard | Provides chemical identifiers, properties, and links to bioassay data [22]. | Used to cross-reference and enrich chemical information retrieved from ECOTOX searches [4] [21]. |
| ECOTOX CTX API | Programmatic interface for querying and retrieving data [23]. | Enables automation of data retrieval for large-scale analysis, model training, and integration into custom research applications. |
| Abstract Sifter | An Excel-based tool for relevance-ranking and triaging PubMed literature search results [22]. | Supports the literature review and curation phase that feeds the quarterly update cycle by efficiently identifying potentially relevant studies. |
| ToxValDB | A compiled database of in vivo toxicology data and derived toxicity values [22]. | Provides a complementary source of mammalian and ecological toxicity data for comparative assessments or weight-of-evidence analyses. |
R Statistical Software with Ecotoxicology Packages (e.g., drc, ssdtools) |
Open-source platform for statistical analysis, including dose-response modeling and species sensitivity distribution fitting [25]. | The primary tool for statistically analyzing endpoint data (e.g., LC50) downloaded from ECOTOX, supporting modern methods like benchmark dose modeling [25]. |
Impact and Applications of Stage 4 Outputs
The effective execution of Stage 4 processes directly enables ECOTOX to fulfill its mission as a critical resource for environmental science and policy.
Support for Regulatory Risk Assessment: ECOTOX is mandated for use in pesticide registration review and ecological risk assessments under statutes like the Clean Water Act and TSCA [4] [6]. The quarterly updates ensure regulators have access to the most recent science. The database's structure directly informs chemical safety evaluations under evolving regulatory frameworks like the EU's REACH 2.0 [26].
Enabling Predictive Toxicology and NAMs: The reliable, structured data from ECOTOX is the empirical bedrock for developing and validating New Approach Methodologies (NAMs). It is used to train QSAR models, machine learning algorithms (as in the ADORE dataset), and to anchor in vitro to in vivo extrapolations [20] [21]. This supports the global regulatory shift toward reducing animal testing [26] [25].
Facilitating Meta-Analysis and Research Synthesis: Researchers leverage the comprehensive, standardized data for large-scale meta-analyses, identification of data gaps, and systematic investigation of chemical effects across species and ecosystems [20]. The public release mechanism ensures this resource is freely available to the global scientific community.
The following diagram summarizes the broader impact pathway of the curated data released through Stage 4:
Diagram 2: Impact Pathway of Publicly Released ECOTOX Data - This diagram shows how the maintained and updated data from Stage 4 feeds into key application areas, leading to defined scientific and regulatory outcomes.
This technical guide details the practical application of the Search and Explore modules within the Ecotoxicology (ECOTOX) Knowledgebase for conducting environmental and human health risk assessments. Framed within a broader thesis on ECOTOX data curation research, this whitepaper provides researchers, scientists, and drug development professionals with a comprehensive methodology for extracting, processing, and applying curated ecotoxicity data. We cover the foundational data curation pipeline that ensures data quality, offer step-by-step protocols for querying the database via its interactive modules and programmatic tools, and present advanced applications in predictive toxicology and machine learning. The integration of these queried data into New Approach Methodologies (NAMs), Adverse Outcome Pathways (AOPs), and quantitative structure-activity relationship (QSAR) models is emphasized as a critical step toward modernizing evidence-based risk assessment [4] [27] [1].
The ECOTOX Knowledgebase, maintained by the U.S. Environmental Protection Agency (EPA), is the world's largest curated repository of single-chemical ecotoxicity data [1]. It is an indispensable resource for ecological risk assessment, chemical safety evaluation, and regulatory decision-making. For over three decades, its systematic and transparent literature review and data curation processes have compiled data from more than 53,000 references, resulting in over one million test records for more than 12,000 chemicals and 13,000 aquatic and terrestrial species [4] [1].
The transition toward evidence-based toxicology and the ethical push to reduce animal testing have increased reliance on curated historical data and NAMs [27] [1]. Within this paradigm, ECOTOX serves a dual purpose: it provides the primary in vivo toxicity data needed for traditional risk assessments, and it supplies the essential training and validation data for developing in silico models and in vitro-to-in vivo extrapolations [28] [1] [29]. Effective querying of this vast resource via its Search and Explore modules is, therefore, a foundational skill for contemporary toxicological research and hazard assessment [4] [30].
Table 1: Scale and Scope of the ECOTOX Knowledgebase (as of 2025) [4] [1]
| Data Category | Count | Description |
|---|---|---|
| Total Test Records | >1,000,000 | Individual curated toxicity test results. |
| Unique Chemicals | >12,000 | Primarily single, organic chemical stressors. |
| Ecological Species | >13,000 | Aquatic and terrestrial plants and animals. |
| Source References | >53,000 | Peer-reviewed literature and grey sources. |
| Data Updates | Quarterly | Regular addition of new curated data. |
Foundational Context: The ECOTOX Data Curation Pipeline
The utility of the Search and Explore modules is predicated on the quality and consistency of the underlying data, ensured by a rigorous curation pipeline. This process aligns with systematic review methodologies and FAIR data principles (Findable, Accessible, Interoperable, Reusable) [1].
The workflow is a multi-stage filter designed to identify, evaluate, and abstract relevant ecotoxicity studies from the scientific literature. It begins with comprehensive searches of the open and grey literature for chemicals of interest [1]. Identified references are screened at the title/abstract and full-text levels against predefined criteria for applicability (e.g., ecologically relevant species, reported exposure concentration) and acceptability (e.g., documented controls, measurable endpoints) [1]. Data from accepted studies are extracted using well-established controlled vocabularies for species, chemicals, endpoints, and test conditions, which is critical for enabling precise queries in the Knowledgebase [4] [1]. This structured curation transforms heterogeneous literature data into a standardized, interoperable format ready for computational analysis and modeling [28] [29].
A Guide to the Search and Explore Modules
The ECOTOX web interface provides two primary modules for data retrieval: Search for targeted queries and Explore for broad investigation [4] [30].
The Search Module: Targeted Data Retrieval
The Search module is designed for users with specific known parameters. It allows direct querying by:
- Chemical: Search by name, CASRN, or DTXSID, with links to the EPA CompTox Chemicals Dashboard for additional physicochemical and hazard data [4] [22].
- Species: Search by common or scientific name across aquatic and terrestrial taxa.
- Effect: Filter by specific biological endpoints (e.g., mortality, growth, reproduction). Users can refine results using up to 19 parameters, including exposure duration, test location, and effect measurement, and customize output from over 100 data fields [4]. This module is ideal for regulatory scenarios requiring specific toxicity values (e.g., LC50 for a particular chemical-species pair) for risk characterization [4].
The Explore Module: Investigative Data Discovery
The Explore module is optimized for discovery and hypothesis generation when search parameters are not precisely defined [4]. Users can start broad queries by chemical, species, or effect, and then iteratively filter and drill down into results using dynamic facets. Key features include:
- Interactive Data Visualization: Results can be visualized as interactive plots. Users can hover over data points for details, zoom into specific areas, and immediately see the impact of applied filters [4].
- Customizable Outputs: Data subsets can be selected and exported in standardized formats (e.g., CSV) for use in external statistical, modeling, or visualization tools [4] [1].
Programmatic Access: The ECOTOXr R Package
For reproducible research and large-scale data analysis, programmatic access is essential. The ECOTOXr R package enables users to build a local SQLite copy of the database and perform documented, reproducible search and extraction procedures directly in R [31].
This approach formalizes the query process, making it shareable, auditable, and integrable with advanced statistical and machine learning workflows in R [31].
Practical Applications in Risk Assessment
Queried data from ECOTOX feed directly into several critical risk assessment frameworks.
Table 2: Key Risk Assessment Use Cases for ECOTOX Data [4] [27] [28]
| Application | Description | Role of Search/Explore Modules |
|---|---|---|
| Chemical Prioritization & Screening | Identifying chemicals of concern based on potency or data gaps. | Rapid retrieval of lowest effect levels across species and endpoints. |
| Deriving Species Sensitivity Distributions (SSDs) | Statistical models to estimate ecosystem-level safe concentrations. | Extracting all available toxicity data for a chemical across multiple species. |
| Informing Adverse Outcome Pathways (AOPs) | Developing mechanistic frameworks linking molecular initiation to adverse outcomes. | Gathering empirical in vivo evidence for key event relationships across biological levels. |
| Validating New Approach Methodologies (NAMs) | Benchmarking in vitro or in silico model predictions against traditional data. | Curating high-quality in vivo reference data for specific chemical and endpoint combinations. |
| Supporting Read-Across & QSAR Modeling | Filling data gaps for untested chemicals using analogs or computational models. | Providing robust training and test datasets of experimental toxicity values. |
Case Study: Integrating Mode of Action (MoA) for Safer Chemical Design A 2024 study curated MoA data for over 3,300 environmentally relevant chemicals and linked them to effect concentrations harvested from ECOTOX [28]. This integrated dataset allows regulators and scientists to group chemicals by shared MoA, establishing meaningful assessment groups for cumulative risk assessment—a process that begins with precise queries in ECOTOX to gather all relevant toxicity data for the chemical list [28].
Case Study: Predicting Aquatic Toxicity of PPCPs
Researchers used ECOTOX to build a dataset for Pharmaceutical and Personal Care Products (PPCPs) [29]. After querying and downloading raw data, they applied a curation tool (Ecotox-curator) to standardize units, resolve duplicates, and classify toxicity based on a 5 mg/L cut-off. This curated dataset was used to develop a Multitasking QSTR model with >85% predictive accuracy, identifying key structural features driving toxicity [29]. This workflow exemplifies the transition from database query to predictive modeling.
Experimental Protocols for Data Retrieval and Curation
Protocol: Systematic Query and Extraction for Model Development
This protocol is adapted from studies building machine learning models from ECOTOX data [32] [29].
- Define Objective: Specify the target (e.g., honey bee acute oral toxicity, fish LC50) [32].
- Query ECOTOX: Use the Explore module with broad initial filters (e.g., species group, endpoint "LC50" or "EC50"). Apply iterative filtering to narrow results.
- Download Data: Export the full dataset using customizable output options.
- Standardize Units: Convert all effect concentrations to a consistent unit (e.g., mg/L, µg/organism). The
ECOTOXrpackage can assist programmatically [32] [31]. - Resolve Duplicates: For each unique chemical-species pair, calculate a representative value (e.g., median or geometric mean) from multiple test records.
- Add Chemical Identifiers: Use CAS numbers to fetch standardized SMILES strings via PubChem or the CompTox Dashboard [32] [29].
- Apply Toxicity Threshold: Classify data based on regulatory thresholds (e.g., EPA's bee toxicity categories: LD50 < 11 µg/bee = highly toxic) [32].
- Curate Final Set: Remove entries with undefined structures (mixtures, inorganics if desired) and perform quality control.
Protocol: Curating Data for a Mode-of-Action Analysis
This protocol is based on the workflow described by [28].
- Compile Chemical List: Assemble target chemicals from monitoring studies, regulatory lists, or production volumes.
- Batch Query Toxicity: For each chemical, use the Search module to retrieve all test records for three key taxonomic groups: algae, crustaceans, and fish.
- Extract Lowest Effect Values: For each chemical-taxon pair, identify the most sensitive endpoint (lowest EC50/LC50) from the curated ECOTOX results.
- Harvest MoA Data: Systematically search literature and specialized databases (e.g., EPA MOAtox, PPDB) for documented MoA for each chemical.
- Integrate Datasets: Merge the curated toxicity data with MoA classifications, creating a unified dataset for analysis and grouping.
Effectively leveraging ECOTOX requires a suite of complementary tools and resources.
Table 3: Research Reagent Solutions for ECOTOX-Based Risk Assessment
| Tool/Resource | Function | Source/Access |
|---|---|---|
| ECOTOX Knowledgebase | Primary source of curated, searchable ecotoxicity data. | U.S. EPA Website [4] |
| CompTox Chemicals Dashboard | Provides complementary chemical data (structures, properties, hazards) linked from ECOTOX searches. | U.S. EPA [4] [22] |
| ECOTOXr R Package | Enables reproducible, programmatic building and querying of a local ECOTOX database. | CRAN / GitHub [31] |
| Ecotox-curator | A Python-based GUI tool to automate the cleaning, standardization, and duplicate removal of raw ECOTOX downloads. | GitHub [29] |
| CIRpy (Python) | A tool to resolve chemical identifiers (e.g., convert CAS numbers to SMILES). | GitHub [29] |
| RDKit | Open-source cheminformatics toolkit used to standardize molecular structures and generate descriptors for modeling. | RDKit.org |
| Mode of Action (MoA) Databases (e.g., MOAtox, PPDB) | Provide mechanistic data to pair with ECOTOX effect concentrations for advanced grouping and AOP development. | U.S. EPA, University of Hertfordshire [28] |
The Search and Explore modules of the ECOTOX Knowledgebase are vital portals for accessing high-quality, curated ecotoxicity data. Mastering their use—from targeted regulatory queries to broad exploratory data mining—is a core competency for modern risk assessors and toxicological researchers. When combined with a rigorous understanding of the underlying data curation pipeline and supplemented with programmatic tools and computational resources, researchers can efficiently transform raw data into actionable knowledge. This process directly supports the advancement of predictive toxicology, the development of NAMs, and the ultimate goal of producing more robust, mechanistic, and timely chemical risk assessments [27] [1].
Evaluating and Overcoming Challenges in Ecotoxicity Data Curation and Use
The ECOTOXicology knowledgebase (ECOTOX) is a critical resource for ecological risk assessment, aggregating peer-reviewed toxicity data for single chemicals across aquatic and terrestrial species. Its utility, however, is fundamentally predicated on the rigor of its data curation process. Within the U.S. Environmental Protection Agency's (EPA) Office of Pesticide Programs (OPP), ECOTOX serves as the primary search engine for identifying open-literature studies to inform regulatory decisions for pesticides[reference:0]. This dual role—as a public repository and a regulatory tool—necessitates a clear, tiered framework for evaluating study acceptability. Understanding the distinction between acceptance for inclusion in the ECOTOX database and acceptance for direct use in OPP risk assessments is therefore a cornerstone of effective data curation and reliable evidence synthesis. This technical guide dissects these acceptance criteria, providing researchers and assessors with a detailed roadmap for interpreting study validity within this critical regulatory context.
Decoding the Acceptance Frameworks
The evaluation process is governed by a phased approach, where Phase I establishes the foundational acceptance criteria for ECOTOX and the additional screens applied by OPP[reference:1]. Studies are subsequently categorized as: (1) accepted by both ECOTOX and OPP, (2) accepted by ECOTOX but not OPP, (3) rejected by both, or (4) placed in an "Other" category for further consideration[reference:2].
Core Criteria for ECOTOX Database Inclusion
For a study to be coded into the ECOTOX database, it must satisfy five minimum criteria ensuring basic data quality and relevance[reference:3].
Table 1: Minimum Acceptance Criteria for the ECOTOX Database
| Criterion | Description | Rationale |
|---|---|---|
| 1. Single Chemical Exposure | The reported toxic effects must result from exposure to a single, identifiable chemical. | Excludes mixture studies to maintain clarity on substance-specific effects. |
| 2. Ecologically Relevant Species | The test organism must be an aquatic or terrestrial plant or animal. | Ensures ecological relevance of the data. |
| 3. Whole-Organism Biological Effect | There must be a measurable biological effect on a live, whole organism. | Excludes in vitro, cellular, or subcellular studies. |
| 4. Reported Concentration/Dose | A concurrent environmental chemical concentration, dose, or application rate is reported. | Essential for quantitative risk assessment. |
| 5. Explicit Exposure Duration | The duration of exposure is explicitly stated. | Allows for differentiation between acute and chronic effects. |
Additional Screening for OPP Regulatory Use
Studies that pass the ECOTOX criteria are subject to a further screen by OPP to determine their utility in pesticide risk assessments. These additional criteria focus on regulatory relevance, data quality, and verifiability[reference:4].
Table 2: Additional OPP Acceptance Criteria for Regulatory Use
| Criterion | Description | Regulatory Rationale |
|---|---|---|
| 6. Chemical of Concern to OPP | Toxicology information is reported for a pesticide or chemical of regulatory interest to OPP. | Ensures data relevance to the agency's mandate. |
| 7. English Language Publication | The article is published in English. | Practical requirement for review and integration. |
| 8. Full Article | The study is presented as a full article (not merely an abstract or conference proceeding). | Ensures sufficient methodological detail for evaluation. |
| 9. Publicly Available Document | The paper is a publicly available document. | Supports transparency and independent verification. |
| 10. Primary Data Source | The paper is the primary source of the data (not a review or secondary analysis). | Ensures traceability and reduces error propagation. |
| 11. Calculated Endpoint | A quantitative endpoint (e.g., LC50, EC10, NOEC) is reported. | Required for benchmark dose analysis and risk quantification. |
| 12. Acceptable Control | Treatment(s) are compared to an acceptable control group. | Establishes baseline response and validates test system. |
| 13. Study Location Reported | The location of the study (laboratory, field, mesocosm) is reported. | Informs the applicability and reliability of the data. |
| 14. Species Verified | The tested species is reported and its identity verified. | Critical for taxonomic specificity and extrapolation. |
Categorization and Rejection
Studies failing the ECOTOX minimum criteria are rejected from the database. Those accepted by ECOTOX but failing one or more OPP criteria (e.g., a study on a non-pesticide chemical, or lacking a calculated endpoint) are categorized as "acceptable for ECOTOX but not OPP"[reference:5]. Attachment I-C of the guidance provides a detailed list of rejection codes for tracking specific deficiencies[reference:6].
Methodological Foundations: Protocols for Key Ecotoxicity Tests
The acceptance criteria presume studies are conducted using sound, standardized methodologies. Below are detailed protocols for two cornerstone tests frequently encountered in regulatory submissions.
Fish Acute Toxicity Test (OECD Test Guideline 203)
This guideline determines the acute lethal toxicity of chemicals to fish, typically via a 96-hour static, semi-static, or flow-through exposure[reference:7].
Experimental Protocol:
- Test Organisms: Healthy, juvenile or adult fish of a defined species (e.g., Danio rerio, Oncorhynchus mykiss). Species, strain, size, weight, and source must be documented.
- Test System: A series of aquaria or test chambers with controlled temperature, pH, dissolved oxygen, and light cycle.
- Exposure Design: A minimum of five test concentrations arranged in a geometric series (factor ≤ 2.2) and a control. Each concentration and control must contain at least seven fish[reference:8].
- Duration & Observations: 96-hour exposure. Mortality is recorded at 24, 48, 72, and 96 hours. Fish are not fed during the test.
- Endpoint: The median lethal concentration (96-h LC50) is calculated using appropriate statistical methods (e.g., probit analysis, Spearman-Karber). A limit test at 100 mg/L may be performed for substances of low toxicity[reference:9].
- Acceptability Criteria (GLP): Test validity requires control mortality ≤ 10%, stable water quality, and compliance with the guideline's procedural specifications.
Avian Acute Oral Toxicity Test (OCSPP 850.2100 / OECD TG 223)
This test estimates the acute oral toxicity of chemicals to birds, providing an LD50 and the slope of the dose-response curve.
Experimental Protocol:
- Test Organisms: Healthy, young-adult birds of a suitable species (e.g., Northern Bobwhite, Mallard). Age, weight, sex, and source are recorded.
- Dosing: The test substance is administered via oral gavage or in a capsule. A vehicle control group is included.
- Experimental Design: Typically, five dose levels are used, with 8-10 birds per dose group. Doses are selected based on range-finding studies.
- Duration & Observations: 14-day observation period post-dosing. Birds are observed daily for mortality, clinical signs of toxicity, and changes in body weight.
- Endpoint: The median lethal dose (LD50) and its 95% confidence interval are calculated using probit or logistic regression analysis. The slope of the dose-response curve is also determined.
- Acceptability Criteria (GLP): Control mortality must be zero, and the study must adhere to Good Laboratory Practice (GLP) standards for data integrity.
Visualizing the Curation and Testing Workflows
Study Acceptance Decision Flowchart
This diagram outlines the logical decision process for categorizing a study based on the ECOTOX and OPP acceptance criteria.
Typical Acute Ecotoxicity Test Workflow
This diagram illustrates the generalized experimental workflow for a standard acute toxicity test, such as the Fish Acute Toxicity Test.
The Scientist's Toolkit: Essential Materials for Ecotoxicity Testing
Conducting studies that meet acceptance criteria requires specific reagents, materials, and tools. The following table details key components of a robust ecotoxicity testing platform.
Table 3: Essential Research Reagent Solutions and Materials
| Item Category | Specific Example(s) | Function in Experiment |
|---|---|---|
| Test Substance | High-purity chemical standard; formulated pesticide product. | The agent whose toxicity is being evaluated. Purity must be characterized. |
| Test Organisms | Defined species of fish (e.g., Danio rerio), birds (e.g., Colinus virginianus), aquatic invertebrates (e.g., Daphnia magna), or plants (e.g., Lemna minor). | The biological model for assessing toxic effects. Must be from a reputable, consistent source. |
| Control Materials | Vehicle control (e.g., solvent, carrier); negative control (clean water, feed). | Establishes baseline organism health and response, validating the test system. |
| Exposure System | Aquaria, flow-through diluters, climate-controlled chambers, oral gavage needles. | Provides the controlled environment for administering the test substance. |
| Water/Air Quality Tools | Dissolved oxygen meter, pH meter, conductivity meter, temperature loggers, ammonia test kits. | Monitors and maintains critical abiotic parameters to ensure test validity. |
| Endpoint Measurement | Mortality records, weighing scales, spectrophotometer (for algal growth), behavioral tracking software. | Quantifies the biological effect (lethal or sublethal) for dose-response analysis. |
| Analytical Chemistry | HPLC, GC-MS, spectrophotometer for chemical analysis of exposure concentrations. | Verifies the actual concentration of the test substance in the exposure medium (TK analysis). |
| Data Analysis Software | Statistical packages (e.g., R, SAS, GraphPad Prism) for probit/logit analysis, ANOVA, LC50/LD50 calculation. | Performs the statistical computations required to generate quantitative endpoints and assess significance. |
The tiered acceptance framework for ECOTOX and OPP is not merely a bureaucratic checkpoint but a fundamental quality assurance mechanism. It ensures that the ECOTOX knowledgebase remains a repository of mechanistically clear, ecologically relevant data, while simultaneously providing OPP risk assessors with a pre-screened, high-utility subset of studies for regulatory decision-making. For researchers, designing studies with these criteria in mind—from employing standardized test guidelines to reporting complete methodological and quantitative data—dramatically increases the likelihood that their work will be accepted and influential. For curators and assessors, a precise understanding of these criteria enables consistent, transparent, and defensible evaluations, ultimately strengthening the scientific foundation of ecological risk assessment. This interpretative clarity is essential for advancing the broader thesis of robust, reliable, and reproducible data curation within environmental toxicology.
Within the rigorous data curation pipeline of the ECOTOXicology Knowledgebase (ECOTOX), the systematic handling of studies with incomplete data or unverified species represents a critical challenge. This in-depth technical guide examines these common pitfalls, framing them within the broader context of ECOTOX's mission to provide reliable, curated ecotoxicity data for environmental research and risk assessment. We detail the operational definitions, identification protocols, and consequential impacts of these data quality issues, providing researchers and curators with explicit methodologies to mitigate their effects and enhance the robustness of ecological toxicity databases.
The ECOTOX Knowledgebase is the world's largest compilation of curated ecotoxicity data, containing over one million test records from more than 13,000 aquatic and terrestrial species and 12,000 chemicals[reference:0]. Its value as a reliable source for chemical assessments hinges on a transparent, systematic review and data curation process designed to identify "relevant and acceptable toxicity results" from the scientific literature[reference:1]. A cornerstone of this process is the application of strict inclusion criteria, which mandate, among other things, "verifiable species" and complete methodological reporting[reference:2]. This guide delves into the practical challenges of upholding these standards, focusing on the pitfalls associated with incomplete data and unverified taxonomic information.
Pitfall 1: Incomplete Data in Ecotoxicity Studies
Definition and Scope
Incomplete data refers to the absence of critical methodological details or results necessary to interpret, replicate, or validate a toxicity test. Within the ECOTOX framework, minimum data requirements for applicability include a quantified chemical exposure, reported duration, a biological response, and basic study quality metrics[reference:3].
Common Manifestations and Identification
The ECOTOX data extraction process captures up to 300–400 data fields per study[reference:4]. Incompleteness can occur in any category, most critically in:
- Chemical Characterization: Missing Chemical Abstracts Service Registry Number (CASRN), purity, or verification of measured vs. nominal concentrations[reference:5].
- Experimental Design: Lack of documented control treatments, exposure media parameters (e.g., pH, temperature), or test method guidelines[reference:6].
- Result Reporting: Endpoints (e.g., LC50, NOEC) reported without associated variance measures, statistical significance, or raw dose-response data.
The ECOTOX screening process identifies these gaps during full-text review, tagging references with exclusion reasons such as 'chemical methods' or insufficient reporting[reference:7].
Impact on Data Utility and Risk Assessment
The use of incomplete data introduces significant uncertainty:
- Impaired Meta-Analysis: Gaps hinder quantitative synthesis and the development of species sensitivity distributions (SSDs).
- Reduced Reliability: Studies lacking control data or chemical verification fail fundamental reliability criteria, potentially leading to erroneous toxicity thresholds.
- Propagation of Data Gaps: ECOTOX may use data from laboratory species to fill gaps for wild species when necessary[reference:8], a practice that can introduce bias if ecological relevance is not carefully considered.
Mitigation Strategies and Curation Protocols
- Proactive Screening: Implement the ECOTOX tiered screening system (title/abstract → full-text) using a predefined checklist based on inclusion criteria (Table 1)[reference:9].
- Transparent Annotation: Clearly flag extracted records with qualifiers indicating missing information (e.g., "nominal concentration assumed," "control not specified") to inform downstream users.
- Strategic Exclusion: Adhere to SOPs that exclude studies missing mandatory fields (e.g., verifiable chemical, control group) while documenting the reason for future potential re-evaluation[reference:10].
Table 1: ECOTOX Inclusion Criteria for Data Completeness (Adapted from ECOTOX SOP)[reference:11]
| Category | Minimum Data Requirement for Inclusion | Common Pitfall (Incomplete Data) |
|---|---|---|
| Chemical (Exposure) | Verifiable CASRN; reported concentration/dose and duration; single-chemical exposure. | Use of trade names only; unreported exposure concentration; missing duration. |
| Species (Population) | Scientific name verifiable against taxonomic sources; life stage information. | Vernacular names only; unspecified organism source or life stage. |
| Comparator | Documented concurrent control treatment (vehicle-only or untreated). | Lack of control description; use of historical controls without justification. |
| Outcome | Biological effect measurement concurrent with exposure; reported endpoint (e.g., LC50, NOEC). | Effect described qualitatively only; endpoint reported without associated data (e.g., confidence limits). |
| Study Reporting | Primary source, full article in English; not a review or abstract only. | Data sourced from secondary summaries or non-peer-reviewed reports without primary reference. |
Pitfall 2: Unverified or Inaccurately Identified Species
The Verification Imperative
ECOTOX requires that "organism taxonomic information [be] verifiable against standard taxonomic sources"[reference:12]. An unverified species is one whose scientific name cannot be confidently resolved to a currently accepted taxon using authoritative references like the Integrated Taxonomic Information System (ITIS) or the Catalogue of Life.
- Use of Synonyms or Obsolete Names: Older studies may use taxonomic names that have since been revised.
- Misidentification: Field-collected organisms may be incorrectly identified by non-specialists.
- Incomplete Specification: Reporting only a genus name (e.g., "Daphnia sp.") or a common name ("fathead minnow") without a definitive scientific identifier.
Consequences for Ecological Relevance
Incorrect species identification severs the link between toxicity data and biology:
- Compromised Cross-Species Extrapolation: Hazard assessments often rely on sensitivity patterns across phylogenetically related species. Misidentification corrupts these models.
- Loss of Ecological Specificity: Data attributed to the wrong species misinforms risk assessments for particular ecosystems or protected species.
- Database Integrity Erosion: Accumulation of unverified records reduces the overall reliability and interoperability of the knowledgebase.
Standard Operating Protocol for Species Verification
The ECOTOX pipeline includes a dedicated SOP for species verification and entry[reference:13]. A practical protocol for curators involves:
- Extraction: Capture the full organism description from the study, including scientific name, authority, life stage, age, and source[reference:14].
- Resolution: Query the provided scientific name against a curated taxonomic backbone (e.g., ITIS API, WORMS for aquatic species).
- Validation: Confirm the name is "accepted" and map any provided synonyms to the current accepted name. Record the taxonomic hierarchy (Kingdom, Phylum, Class, Order, Family, Genus, Species).
- Flagging and Action:
- Verified: Enter with full taxonomy.
- Unverified (Unresolvable): Flag the record. Depending on project needs, the study may be excluded or entered with a warning annotation.
- Ambiguous (e.g., "sp."): Enter at the lowest verifiable taxonomic level (e.g., genus) and flag the record's limited specificity.
Table 2: Impact of Data Completeness and Species Verification on Study Inclusion
| Data Quality Scenario | ECOTOX Screening Decision | Rationale & Curatorial Action |
|---|---|---|
| Complete data, verified species | Include | Meets all applicability and acceptability criteria. Data extracted into all relevant fields. |
| Complete data, unverified species | Typically Exclude | Fails mandatory requirement for verifiable taxonomic information. Tagged with exclusion reason. |
| Incomplete critical data (e.g., no control), verified species | Exclude | Fails acceptability criteria. Documented control is required for inclusion[reference:15]. |
| Partial data (e.g., missing pH), verified species | Conditional Include | May be included if core requirements (chemical, species, endpoint, control) are met. Missing parameters flagged. |
Experimental Protocol: Benchmarking Data Quality
To operationalize the identification of incomplete data, a standardized audit protocol can be applied to a dataset. The following methodology is adapted from ECOTOX's systematic review principles.
Protocol: Quality Audit of Ecotoxicity Data Extracts
Objective: To quantify the frequency and typology of data incompleteness in a set of ecotoxicity study records.
Materials: A sample of extracted data records (e.g., 100 records from ECOTOX); a checklist of mandatory and desirable data fields derived from ECOTOX's 300+ field schema[reference:16]; a taxonomic verification tool (e.g., ITIS web service).
Procedure:
- Field Completeness Audit: For each record, score each mandatory field (e.g., CASRN, species binomial, endpoint value, control type) as "Complete," "Partial," or "Missing."
- Taxonomic Verification: For the species name in each record, submit the name to the verification tool. Record the outcome as "Accepted," "Synonym (Mapped to Accepted)," or "Unresolved."
- Source Traceability: Verify that the cited reference is a primary, full-text article.
- Statistical Analysis: Calculate the percentage of records with missing mandatory fields and the percentage with unverified species. Categorize incompleteness by data module (Chemical, Species, Test Conditions, Results).
Expected Output: A quantitative profile of data gaps, informing priorities for curation refinement, targeted literature re-extraction, or guidance for primary researchers.
Visualizing the Curation Pipeline and Decision Logic
Diagram 1: ECOTOX Data Curation and Quality Screening Pipeline
This diagram outlines the key stages of the ECOTOX literature review and data curation pipeline, highlighting points where checks for data completeness and species verification occur[reference:17].
Diagram 2: Decision Logic for Handling Incomplete Data & Unverified Species
This flowchart illustrates the curatorial decision-making process when encountering studies with incomplete data or unverified species during the ECOTOX full-text review and data extraction stages.
The Scientist's Toolkit: Essential Reagents & Materials for Curated Ecotoxicity
This table details key materials and resources essential for conducting high-quality ecotoxicity tests that would meet ECOTOX inclusion criteria, thereby avoiding the pitfalls of incomplete data.
Table 3: Research Reagent Solutions for Robust Ecotoxicity Testing
| Item Category | Specific Item / Resource | Function in Experiment | Relevance to Avoiding Pitfalls |
|---|---|---|---|
| Test Organism | Certified culture of Daphnia magna (e.g., EPA clone) | Standardized, sensitive freshwater invertebrate for acute/chronic tests. | Use of a well-defined, traceable species source ensures taxonomic verification and reproducibility. |
| Chemical Standard | Analytical standard of target chemical with known CASRN and purity (>98%). | Provides exact identity and concentration for exposure solutions. | Enables complete chemical characterization (CASRN, purity) and accurate dose reporting. |
| Control Reagent | Certified solvent (e.g., acetone, DMSO) for vehicle control. | Establishes a baseline for effects not attributable to the test chemical. | Mandatory for meeting ECOTOX acceptability criteria requiring a documented control[reference:18]. |
| Water Quality | Multiparameter probe (pH, dissolved O₂, conductivity, temperature). | Monitors and maintains optimal and stable test conditions. | Allows complete reporting of test conditions, a key data field often incomplete. |
| Taxonomic Reference | Integrated Taxonomic Information System (ITIS) web service. | Authoritative source for verifying scientific names and taxonomic hierarchy. | Critical tool for curators and researchers to ensure species verification prior to publication. |
| Test Guideline | OECD Test Guideline 203 (Fish, Acute Toxicity Test)[reference:19]. | Provides internationally recognized protocol for study design and reporting. | Following standardized guidelines inherently improves data completeness and reliability. |
Navigating the pitfalls of incomplete data and unverified species is not merely a technical curatorial task but a fundamental component of maintaining the scientific integrity of ecological toxicity databases like ECOTOX. By adhering to explicit, transparent protocols for data screening, verification, and extraction—and by employing the tools and guidelines outlined herein—researchers and curators can significantly enhance the reliability and utility of ecotoxicity data. This, in turn, strengthens the foundation of evidence-based environmental risk assessment and chemical safety decision-making. The ongoing evolution of the ECOTOX Knowledgebase demonstrates that rigorous, systematic handling of these pitfalls is achievable and essential for supporting future research and policy.
The Ecotoxicology (ECOTOX) Knowledgebase serves as a pivotal, authoritative source for curated single-chemical toxicity data, supporting environmental research, chemical risk assessments, and the development of predictive models [4]. As a comprehensive, publicly available resource compiled from over 53,000 scientific references, it contains more than one million test records covering over 13,000 species and 12,000 chemicals [4] [20]. Its primary function is to provide systematically reviewed ecotoxicity data that informs regulatory mandates under acts like the Clean Water Act and the Toxic Substances Control Act (TSCA) [4] [20].
Within the context of a broader thesis on ECOTOX knowledgebase data curation process research, this guide addresses a critical, nuanced component: the management of studies that do not fit standard evaluation criteria and thus require additional expert judgment. The shift towards New Approach Methodologies (NAMs) and computational toxicology increases the value of curated in vivo data for validation but also introduces more complex study designs that challenge traditional curation workflows [20]. This document provides a technical framework for identifying, processing, and integrating these "Other" category papers, ensuring the continued reliability and FAIRness (Findable, Accessible, Interoperable, and Reusable) of the knowledgebase [20] [5].
Defining the 'Other' Category in Data Curation
In the ECOTOX data curation pipeline, the 'Other' category is not a repository for low-quality studies but a classification for scientifically sound papers that present unique complexities precluding straightforward classification using standard controlled vocabularies and quality scoring systems. The classification of a study into this category necessitates additional expert judgment to determine its ultimate utility and integration path.
The table below outlines the primary criteria and specific examples that trigger assignment to this category.
Table: Criteria for Assigning Studies to the 'Other' Category
| Criterion Category | Specific Triggers | Examples from Ecotoxicology Literature |
|---|---|---|
| Non-Standard Endpoints | Measurement of biological effects not captured by standard ECOTOX endpoint vocabularies (e.g., "mortality," "growth," "reproduction"). | Transcriptomic changes, metabolomic profiles, novel behavioral assays, histological alterations not linked to a standard apical endpoint. |
| Complex Experimental Designs | Studies that deviate from standard single-species, constant-exposure tests. | Multi-generational studies, multi-stressor experiments (e.g., chemical + temperature), field mesocosm studies with numerous species. |
| Emerging Contaminants & Classes | Data on chemical classes with poorly defined modes of action or for which assessment frameworks are still under development. | Studies on per- and polyfluoroalkyl substances (PFAS), complex polymer degradation products, or engineered nanomaterials [33]. |
| Ambiguous or Incomplete Reporting | Studies where key methodological details are omitted but the core data may still be valuable. | Lack of explicit concentration units, unspecified exposure duration, or use of a non-standard test species without adequate taxonomic detail. |
The process for handling these studies is formalized to minimize subjectivity. As outlined in general expert judgment frameworks, it involves clearly defining the problem, documenting specific questions for the expert, and selecting qualified subject matter experts (SMEs) with deep knowledge in the relevant domain (e.g., avian toxicology, sediment chemistry, computational biology) [34]. In the ECOTOX context, this often involves internal curation team leads or designated external consultants.
The ECOTOX curation process follows a systematic review framework to ensure transparency, objectivity, and consistency [20]. The protocol for handling papers in the 'Other' category integrates directly into this established workflow. The following diagram illustrates the key decision points and integration of expert judgment.
Diagram: ECOTOX Curation Workflow with Expert Judgment Pathway. This shows the integration point for 'Other' category papers.
Detailed Methodology
Literature Search and Acquisition: Potential studies are identified through comprehensive, automated searches of scientific databases using predefined search strings for ecotoxicology terms, combined with manual monitoring of key journals [20].
Initial Screening and Extraction: Trained curators perform an initial review, extracting core data (chemical, species, endpoint, effect value) using controlled vocabularies. At this stage, papers with obvious non-standard elements are flagged.
Quality Assessment & 'Other' Flagging: Each study undergoes a quality evaluation, often based on criteria similar to the Klimisch score, which categorizes studies based on reliability (e.g., 1=reliable without restriction, 4=not assignable) [20]. Studies deemed scientifically sound (Klimisch 1 or 2) but which cannot be fully categorized due to the triggers in Section 2 are formally assigned to the 'Other' category.
Structured Expert Elicitation:
- Question Formulation: For each flagged paper, the lead curator drafts a specific set of questions. Example: "Does the measured 'oxidative stress gene cluster upregulation' in this study correspond to a traditional apical endpoint like 'histopathology' or 'survival' for the purpose of benchmark dose modeling?"
- Expert Selection: An SME is chosen based on the paper's topic (e.g., a molecular toxicologist for a transcriptomics study).
- Elicitation and Documentation: The expert reviews the paper and questions. Their judgment, along with a clear rationale, is recorded in a standardized form within the curation tracking system. This process aligns with steps outlined for formal expert judgment, which involves submitting questions, reviewing judgments, and aggregating them into a report [34].
Final Data Integration: Based on the expert's decision, the paper is either excluded, or its data is curated with custom annotations or fields that capture the non-standard information, making it accessible for advanced queries and interoperable with tools like the CompTox Chemicals Dashboard [22] [20].
Case Studies: Applying Expert Judgment
Case Study 1: Curating Data for PFAS Compounds
Per- and polyfluoroalkyl substances (PFAS) represent a major class of 'Other' category triggers due to their complex chemistry, environmental persistence, and diverse, often poorly characterized modes of action [33]. A study investigating the subcellular histological effects of perfluorooctanesulfonic acid (PFOS) on fish liver may report endpoints like "peroxisome proliferation" not found in standard lists.
- Expert Judgment Elicited: A toxicologist with expertise in PFAS mechanisms and fish histopathology was consulted.
- Key Questions:
- Is peroxisome proliferation a consistent and relevant adverse outcome for PFAS in this species?
- Can the quantitative data (e.g., proliferation incidence) be reasonably mapped to a traditional endpoint like "organ pathology" for regulatory benchmark derivation?
- Outcome: The expert affirmed the relevance and provided a scaling factor to relate the histological score to a categorical pathology severity. The data was curated under the endpoint "histopathology - liver" with a special annotation detailing the expert's rationale and the original measurement, preserving both the standard and specific information.
Case Study 2: Interpreting a High-Throughput Transcriptomics (HTTr) Study
A paper using High-Throughput Transcriptomics (HTTr) to assess the effects of a novel pesticide on a cell line presents concentration-response data for hundreds of gene pathways [22]. This is a core New Approach Methodology (NAM).
- Expert Judgment Elicited: A computational toxicologist specializing in toxicogenomics and adverse outcome pathway (AOP) development.
- Key Questions:
- Which of the significantly altered gene pathways are linked to established AOPs relevant to ecologically significant apical endpoints (e.g., impaired reproduction, growth)?
- What is the most appropriate method to derive a point-of-departure (e.g., AC50) from this multidimensional data for use in a quantitative in vitro to in vivo extrapolation (QIVIVE) model?
- Outcome: The expert identified three key pathways linked to vertebrate estrogen signaling. They recommended a specific bioinformatics pipeline (e.g., using ToxCast pipeline data [22]) to calculate an overall bioactivity score. The curated record includes the derived AC50, the list of key pathways, and a link to the relevant AOP-Wiki identifiers, enabling interoperability with computational toxicology resources.
Table: Summary of Case Study Outcomes
| Case Study | 'Other' Category Trigger | Expert Domain | Core Judgment | Integration into ECOTOX |
|---|---|---|---|---|
| PFAS Histology | Non-standard endpoint (peroxisome proliferation) | PFAS Toxicologist / Histopathologist | Mapping of subcellular change to adverse organ-level pathology. | Data curated under standard "histopathology" with detailed expert annotation. |
| HTTr Pesticide | Complex NAM data (transcriptomic pathways) | Computational Toxicologist / AOP Developer | Identification of relevant AOP-linked pathways & derivation of bioactivity score. | Point-of-departure value stored with links to AOP and assay metadata. |
Effectively navigating the 'Other' category and leveraging the broader ECOTOX database requires a suite of specialized tools and resources. The following table details key components of this toolkit.
Table: Research Reagent Solutions for ECOTOX Data Curation and Analysis
| Tool/Resource | Function/Benefit | Relevance to 'Other' Category |
|---|---|---|
| ECOTOXr R Package [5] | Enables reproducible, programmatic access to the ECOTOX database via R scripts. Formalizes data retrieval, filtering, and analysis, ensuring transparency and reproducibility for meta-analyses. | Allows researchers to systematically retrieve and analyze studies that may have non-standard annotations, facilitating the batch processing of 'Other' category data for model development. |
| Klimisch Scoring System | A standardized reliability assessment framework for toxicological studies. Helps categorize studies as "reliable," "reliable with restrictions," "not reliable," or "not assignable." | Provides the initial quality filter. 'Other' category papers are often "reliable with restrictions" (Klimisch 2), where expert judgment resolves the restriction. |
| CompTox Chemicals Dashboard [4] [22] | A hub for chemistry, toxicity, and exposure data for thousands of chemicals. Integrates with ECOTOX, providing chemical identifiers, properties, and links to ToxCast assay data. | Critical for contextualizing chemicals from 'Other' category studies, especially emerging ones like PFAS, by linking them to molecular structures and high-throughput screening data. |
| Abstract Sifter [22] | An Excel-based tool that enhances literature triage in PubMed. It uses text mining to rank abstracts by relevance to a user-defined set of keywords (e.g., chemical names, endpoints). | Accelerates the initial identification phase of the systematic review process, helping curators efficiently find papers that may eventually require expert judgment. |
| ToxValDB (v9.6+) [22] | A large compilation of summary toxicology values (e.g., benchmark doses) from multiple sources, standardized for comparison. | Serves as a key reference for experts when deciding how to quantify effects from 'Other' category papers for use in derived value calculations. |
| High-Throughput Toxicokinetics (HTTK) Data [22] | Provides in vitro toxicokinetic parameters for hundreds of chemicals, enabling the prediction of in vivo blood/tissue concentrations from in vitro assay data. | Essential for interpreting 'Other' category NAM studies (e.g., cell-based HTTr) by providing the means to perform quantitative in vitro to in vivo extrapolation (QIVIVE). |
The 'Other' category is an essential, dynamic component of a robust ecotoxicology data curation framework. It ensures that valuable, cutting-edge science is not excluded due to the inherent lag between scientific innovation and the development of standardized data schemas. The formal integration of structured expert judgment transforms this category from a holding bin into a critical pathway for knowledgebase evolution and relevance.
Future advancements in this area will likely focus on increasing automation and reducing expert burden. This could involve:
- Developing machine learning classifiers trained on past expert decisions to pre-flag and suggest categorizations for new 'Other' studies.
- Creating more granular and flexible controlled vocabularies that can be extended by the expert community, particularly for NAM endpoints and emerging contaminant classes.
- Enhancing the interoperability of expert annotations so that the rationale behind curating a complex PFAS or transcriptomics study is seamlessly viewable and computable across linked platforms like the CompTox Dashboard and AOP-KB.
By maintaining rigorous, documented protocols for expert elicitation as detailed in this guide, the ECOTOX Knowledgebase can continue to fulfill its mission as a FAIR, authoritative resource that supports both regulatory decision-making and scientific discovery in an era of rapidly evolving toxicological science.
The exponential growth of chemicals in commerce has created an urgent need for rapid, reliable, and efficient methods for ecological hazard assessment [1]. In this context, curated databases are not merely repositories but foundational tools for scientific research and regulatory decision-making. The ECOTOX Knowledgebase stands as the world's largest compilation of curated single-chemical ecotoxicity data, serving as a critical resource for researchers and risk assessors [1]. Concurrently, the CompTox Chemicals Dashboard provides a complementary platform integrating chemistry, exposure, and toxicity data for over a million chemical substances [11]. The interoperability between these systems represents a paradigm shift in how environmental scientists access and utilize data.
Framed within broader research on data curation processes, this guide examines the technical methodologies for optimizing data retrieval. The curation pipeline of ECOTOX itself is a rigorous exercise in systematic review, employing transparent literature search, study evaluation, and data abstraction protocols that align with modern FAIR (Findable, Accessible, Interoperable, Reusable) data principles [1]. The value of this meticulously curated data is fully realized only when researchers can effectively filter, visualize, and connect it to other computational resources. This guide provides an in-depth technical exploration of these optimization techniques, detailing how strategic use of search filters, advanced visualizations, and tool interoperability can accelerate hypothesis generation, chemical prioritization, and ecological risk assessment.
Understanding the technical capabilities and content scope of the ECOTOX Knowledgebase and the CompTox Chemicals Dashboard is essential for designing effective search strategies. The following table compares the core attributes of these two interoperable platforms.
Table 1: Comparative Scope and Content of ECOTOX Knowledgebase and CompTox Chemicals Dashboard
| Feature | ECOTOX Knowledgebase | CompTox Chemicals Dashboard |
|---|---|---|
| Primary Focus | Curated ecotoxicity effects data for ecological species [4]. | Integrated chemistry, toxicity, and exposure data for chemical substances [11]. |
| Key Data Types | Test conditions, measured effects (mortality, growth, reproduction), endpoints (LC50, NOEC), species details [4] [1]. | Chemical identifiers, structures, physicochemical properties, predicted and experimental hazard data, bioassay results, product use, exposure estimates [11] [35]. |
| Data Volume | >1 million test records [4]. | >1 million chemical substances [11]. |
| Coverage | >12,000 chemicals, >13,000 aquatic & terrestrial species [4]. | Extensive chemical space with over 300 curated chemical lists (e.g., pesticides, PFAS) [11]. |
| Source | Peer-reviewed literature, systematically curated [1]. | EPA databases, PubChem, ECOTOX, and other public sources [11]. |
| Core Function | Answer: "What are the toxic effects of this chemical on ecological organisms?" | Answer: "What are the properties of this chemical, and what is known about its hazard and exposure?" |
The ECOTOX Knowledgebase is built on a systematic data curation pipeline. The process begins with comprehensive literature searches, followed by a tiered review of titles, abstracts, and full texts against predefined criteria for applicability and acceptability [1]. Pertinent methodological details and results are then extracted using controlled vocabularies to ensure consistency. This rigorous process, detailed in the table below, ensures the reliability of the over one million bioassay records available for querying [1].
Table 2: ECOTOX Systematic Data Curation Protocol
| Curation Phase | Key Activities | Quality Control / Output |
|---|---|---|
| Literature Search & Acquisition | Development of chemical-specific search strings; searching of bibliographic databases (e.g., PubMed, Scopus) and grey literature [1]. | Comprehensive reference library for target chemicals. |
| Citation Screening | Title/abstract review against applicability criteria (ecologically relevant species, single chemical, defined exposure); full-text review for acceptability criteria (documented controls, reported endpoint) [1]. | PRISMA-style flow diagram of included/excluded studies [1]. |
| Data Abstraction | Extraction of ~100 data fields into standardized forms: chemical, species, test design, conditions, results (e.g., effect concentration, statistical significance) [1]. | Curated record with controlled vocabulary (e.g., specific endpoint terms like "LC50"). |
| Data Maintenance & Release | Quarterly updates with new data; resolution of user feedback; quality assurance checks [4] [1]. | Public release of validated data via web interface and API. |
Optimizing Searches: Strategic Use of Filters and Taxonomies
Efficient data retrieval from large-scale repositories like ECOTOX and the CompTox Dashboard requires moving beyond basic keyword searches. Mastery of their structured filtering systems is key to precision.
1. ECOTOX Search Refinement: The ECOTOX interface allows searches to be initiated by Chemical, Species, or Effect [4]. The true power lies in the ability to refine results using up to 19 parameters, including Exposure Duration, Endpoint (e.g., mortality, growth), Effect (e.g., lethal, sublethal), Test Location (field vs. lab), and Publication Year [4]. For example, a search for the chemical "chlorpyrifos" can be quickly narrowed to only chronic toxicity studies (Exposure Duration > 10 days) on freshwater fish species, yielding the most relevant data for a long-term risk assessment.
2. CompTox Dashboard Filtering Taxonomy: The Dashboard employs a multi-layered filtering taxonomy [35]. Users can pre-filter searches to exclude isotopically labeled compounds or multi-component substances. Post-search, the extensive "Table View" allows filtering by data availability—such as the presence of toxicity values, bioassay results, or physicochemical properties—enabling rapid identification of data-rich versus data-poor chemicals for testing prioritization [35].
3. Leveraging Chemical Lists: A powerful feature for regulatory and investigative work is the use of curated chemical lists. The Dashboard contains over 300 lists categorized by structure (e.g., per- and polyfluoroalkyl substances - PFAS), use (e.g., pesticides, antimicrobials), or regulatory status [11] [35]. A researcher can filter the entire Dashboard to show only chemicals on the "EPA PFAS Master List," immediately focusing their analysis on this critical class.
Table 3: Search Filtering Taxonomies for Targeted Data Retrieval
| Tool | Filter Category | Example Filters | Use Case |
|---|---|---|---|
| ECOTOX [4] | Study Design | Exposure duration, Test location (field/lab), Route of exposure, Test medium (water, sediment). | Find chronic, water-only laboratory studies for criterion derivation. |
| Biological System | Species, Species group (e.g., fish, amphibian), Life stage, Sex. | Extract data for a sensitive keystone species (e.g., Daphnia magna). | |
| Measured Outcome | Endpoint (LC50, NOEC, EC50), Effect (mortality, reproduction, behavior). | Compare acute lethality (LC50) across taxa for a chemical. | |
| CompTox Dashboard [11] [35] | Data Availability | Presence of: ToxCast assay data, physicochemical properties, hazard data, exposure data. | Identify chemicals with sufficient data for model building vs. those with data gaps. |
| Chemical Identity | Chemical list membership, Structure-based category, Single-component vs. mixture. | Investigate the properties of all chemicals in a specific regulatory list (e.g., TSCA Active Inventory). | |
| Property Range | Molecular weight, Log P (octanol-water coefficient), Water solubility. | Screen for chemicals with properties indicating high environmental mobility or bioaccumulation potential. |
From Data to Insight: Advanced Visualization and Analysis Techniques
Raw data tables are often insufficient for pattern recognition. Both ECOTOX and the broader computational toxicology ecosystem provide visualization tools to transform data into insight.
ECOTOX Integrated Visualizations: Within ECOTOX, the Data Visualization feature generates interactive plots of search results [4]. A user can plot effect concentrations (e.g., LC50) against exposure duration or visualize the distribution of sensitivity across different species groups. These interactive charts allow users to hover over data points to reveal underlying study details, facilitating quick exploration and identification of outliers or trends [4].
The ToxPi Framework for Integrative Profiling: For a higher-level, multi-attribute comparison of chemicals, the Toxicological Prioritization Index (ToxPi) framework is indispensable [36]. ToxPi integrates and normalizes disparate data streams (e.g., hazard scores, exposure potential, physicochemical properties) into a single visual profile shaped like a pie chart. Each "slice" represents a different data domain, and its size indicates the relative contribution to the overall concern score [36]. This allows researchers to visually compare dozens of chemicals and immediately understand which factors (e.g., high hazard, wide exposure) drive priority. The framework is supported by the ToxPi*GIS Toolkit, which enables the creation of geospatial maps where each location displays a ToxPi profile, linking chemical hazard to geography [36].
Interoperability for Pathway Analysis: Advanced analysis often requires exporting data. The CompTox Dashboard facilitates this by providing direct links to related resources and downloadable files in standard formats (SDF, CSV) [35]. A critical link is to the Adverse Outcome Pathway (AOP) Wiki, accessible from a chemical's Executive Summary tab [35]. This allows a researcher viewing a chemical like a pharmaceutical that activates a specific receptor to immediately navigate to the relevant AOP, connecting the chemical to a structured sequence of mechanistic events leading to an adverse ecological outcome. This interoperability between chemical-specific data and pathway knowledge is a cornerstone of modern, mechanism-based risk assessment.
The Scientist's Toolkit: Essential Research Reagent Solutions
Modern ecotoxicology research relies on a suite of digital "reagents" and materials that facilitate the access, analysis, and interpretation of curated data.
Table 4: Key Digital Research Reagent Solutions in Computational Ecotoxicology
| Tool / Resource | Function | Typical Application |
|---|---|---|
| DTXSID (DSSTox Substance Identifier) | A unique, stable identifier for a chemical substance within the EPA CompTox chemistry infrastructure [11] [35]. | The preferred identifier for programmatically linking data across CompTox, ECOTOX, and other EPA tools, ensuring unambiguous chemical referencing. |
| ToxPi Graphical User Interface (GUI) | A stand-alone software application for creating ToxPi models by integrating and weighting diverse data slices [36]. | Building a chemical prioritization index for a set of contaminants in a watershed, combining hazard, use, and detection frequency data. |
| QSAR-ready SMILES | A standardized molecular structure representation prepared for quantitative structure-activity relationship modeling [35]. | Serving as the direct input for computational models (e.g., OPERA) to predict missing physicochemical or toxicity properties for a chemical. |
| ECOTOX API (Application Programming Interface) | A programmatic interface allowing direct querying and retrieval of ECOTOX data by external software [1]. | Automating the extraction of all toxicity data for a list of chemicals into a custom script for species sensitivity distribution (SSD) analysis. |
| SeqAPASS Tool | An online tool that extrapolates toxicity information across species based on sequence similarity of specific protein targets [37]. | Predicting the potential susceptibility of a non-test species (e.g., an endangered fish) to a chemical by comparing its protein targets to those of a well-studied model species. |
| Generalized Read-Across (GenRA) Tool | An algorithmic tool within the CompTox Dashboard that predicts toxicity by identifying and using data from structurally similar chemicals [37]. | Providing a hypothesis-driven, quantitative estimate of chronic toxicity for a data-poor chemical by reading across from well-studied analogues. |
Experimental Protocol: Curating Mode-of-Action Data from ECOTOX for Hazard Assessment
The following protocol details a methodology for harvesting and curating data from ECOTOX to support advanced hazard assessment, specifically for developing mode-of-action (MoA) classifications—a task critical for grouping chemicals and applying new approach methodologies (NAMs) [28].
Objective: To compile a curated dataset of aquatic effect concentrations and associated MoA classifications for environmentally relevant chemicals, using ECOTOX as the primary toxicity data source.
Materials & Data Sources:
- Chemical List: A predefined list of target chemicals (e.g., 3,387 substances from environmental monitoring suspect lists) [28].
- Primary Database: US EPA ECOTOX Knowledgebase (https://www.epa.gov/ecotox).
- MoA Reference Resources: Specialized databases (e.g., EPA MOAtox, PPDB, DrugBank) and scientific literature [28].
- Data Management Software: Spreadsheet or database application (e.g., Microsoft Excel, PostgreSQL) with scripting capability (e.g., Python, R) for data processing.
Procedure: Step 1: Toxicity Data Harvesting from ECOTOX
- For each target chemical, execute a systematic query in the ECOTOX Knowledgebase using the chemical's name or CASRN.
- Apply filters to retrieve data for the three standard aquatic trophic levels: algae, crustaceans (e.g., Daphnia), and fish.
- Further refine results to include key endpoints: EC50 (algae, crustaceans), LC50 (fish), and chronic NOEC/LOEC values where available.
- Use the batch export function to download the full dataset, including metadata on species, test duration, endpoint, and effect concentration.
Step 2: Data Curation and Aggregation
- Clean the Data: Standardize chemical identifiers (prioritize DTXSID), resolve synonyms, and flag salts or mixtures.
- Apply Quality Filters: Exclude records with undefined exposure durations, non-standard endpoints, or studies with significant confounding factors as noted in the ECOTOX record.
- Aggregate by Species Group: For each chemical and species group (algae, crustacea, fish), calculate a geometric mean of all valid effect concentrations (e.g., EC50) to derive a single, representative value for acute toxicity. Document the sample size (number of studies/species) and range.
Step 3: Mode-of-Action (MoA) Research and Classification
- Systematic MoA Search: For each chemical, query multiple MoA reference databases and conduct targeted literature searches using the chemical name combined with terms like "mode of action" and "toxicity" [28].
- Data Synthesis: Collect all MoA information. Classify the primary MoA into broad, standardized categories (e.g., "Acetylcholinesterase inhibition," "Estrogen receptor agonist," "Uncoupling of oxidative phosphorylation") [28].
- Curation and Assignment: Resolve conflicting MoA information by applying predefined criteria (e.g., prioritizing evidence from mammalian or insect target assays for specific receptors). Assign a confidence level (e.g., High, Medium, Low) to the MoA classification based on the quantity and quality of underlying evidence.
Step 4: Dataset Integration and FAIR Formatting
- Merge the curated toxicity data table with the MoA classification table using a stable chemical identifier (DTXSID).
- Structure the final dataset according to FAIR principles: include a data dictionary, provenance information (source databases, query dates), and clear licensing terms.
- Publish the dataset in an open-access repository (e.g., Zenodo, Figshare) in both human-readable (CSV) and machine-actionable (JSON-LD) formats.
The strategic integration of optimized search techniques, advanced visualizations, and tool interoperability moves the field from descriptive data compilation to predictive and mechanistic science. The rigorous curation process of the ECOTOX Knowledgebase provides the essential, high-quality empirical foundation [1]. Tools like the CompTox Dashboard and ToxPi then enable the synthesis of this data with chemical properties and exposure information to form integrated profiles [11] [36].
The future of this ecosystem lies in enhanced automation and artificial intelligence. Machine learning models trained on curated ECOTOX data are already being used to predict toxicity for untested chemicals. Furthermore, the deepening integration with Adverse Outcome Pathway (AOP) networks promises a more fundamental shift. The vision is a fully connected knowledge system where a search for a chemical not only returns toxicity values but also maps those effects onto defined molecular initiating events and key biological pathways, directly informing the use of relevant New Approach Methodologies (NAMs) like high-throughput in vitro assays [1] [28]. By mastering the current technical landscape outlined in this guide, researchers position themselves to lead the development and application of these next-generation, knowledge-driven assessment paradigms.
Validating ECOTOX Data Quality and Comparative Analysis in Real-World Research
The exponential growth of chemical substances in commerce necessitates robust, transparent, and reusable ecological toxicity data for environmental risk assessment and regulatory decision-making. The FAIR (Findable, Accessible, Interoperable, Reusable) principles, established as a guideline to improve the reusability of digital assets, provide a critical framework for achieving this goal[reference:0]. Within the context of ecotoxicology, the U.S. Environmental Protection Agency's (EPA) ECOTOXicology Knowledgebase (ECOTOX) represents a seminal effort to operationalize these principles through a systematic, large-scale data curation process. This technical guide examines the adherence to FAIR principles within the ECOTOX knowledgebase, framing it as a cornerstone case study in the broader thesis on advancing data curation methodologies for ecological risk assessment.
The scale and scope of ECOTOX underpin its utility as a FAIR-aligned resource. The following table summarizes key quantitative metrics that demonstrate its comprehensiveness and ongoing growth.
Table 1: Quantitative Profile of the ECOTOX Knowledgebase (Version 5)
| Metric | Value | Significance |
|---|---|---|
| Curated Toxicity Records | >1,000,000 records | Represents the world's largest compilation of curated single-chemical ecotoxicity test results[reference:1]. |
| Source References | >50,000 references | Indicates extensive coverage of the peer-reviewed and grey literature[reference:2]. |
| Unique Chemicals | >12,000 chemicals | Supports hazard assessment for a vast array of environmental contaminants[reference:3]. |
| Ecological Species Covered | Aquatic & terrestrial plants, invertebrates, vertebrates | Ensures relevance for holistic ecological risk assessments across taxa[reference:4]. |
| Data Curation Pipeline SOPs | 3 core SOPs (Literature Search, Data Abstraction, Data Maintenance) plus supporting SOPs | Standardizes the review process, enhancing transparency and consistency[reference:5]. |
| Update Frequency | Quarterly data additions | Maintains the database as an "evergreen" resource with current science[reference:6]. |
Implementing FAIR Principles in the ECOTOX Data Curation Pipeline
ECOTOX's design and operations are explicitly aligned with FAIR principles, transforming raw literature into a structured, reusable resource[reference:7]. The table below details the practical implementation of each principle.
Table 2: Mapping FAIR Principles to ECOTOX Curation Practices
| FAIR Principle | ECOTOX Implementation | Technical Detail / Standard |
|---|---|---|
| Findable | • Persistent Query Interface: Public web interface (www.epa.gov/ecotox) with advanced search filters.• Rich Metadata: Every record includes structured metadata (chemical CAS RN, species taxonomy, test conditions)[reference:8].• Standardized Vocabularies: Use of controlled terms for effects, endpoints, and test methods. | Metadata fields follow a defined schema (e.g., chemical purity, exposure concentration type) to facilitate discovery by both humans and machines[reference:9]. |
| Accessible | • Open Access: Data is publicly retrievable via the web interface without authentication for most uses.• Standardized Retrieval: The ECOTOXr R package provides programmatic, reproducible access to the database[reference:10].• Clear Usage Policies: Licensing and citation guidelines are provided. |
The ECOTOXr package formalizes data retrieval as a scriptable workflow, ensuring transparent and consistent accessibility for computational reuse[reference:11]. |
| Interoperable | • Structured Data Model: Data is extracted into a consistent relational schema (e.g., separate tables for chemicals, species, results).• Chemical Identifiers: Mandatory use of Chemical Abstracts Service Registry Numbers (CAS RN)[reference:12].• Taxonomic Verification: Species names are verified against standard sources[reference:13].• Tool Integration: Data is used for QSAR modeling, species sensitivity distributions (SSDs), and interoperability with other EPA tools[reference:14]. | The use of canonical identifiers (CAS RN, scientific names) and a consistent data model enables seamless integration with other toxicological databases and computational pipelines. |
| Reusable | • Detailed Provenance: Each record is linked to its source reference, with extracted methodological details (e.g., test duration, control data)[reference:15].• Rich Context: Fields capture study design, test conditions, and statistical significance, allowing for informed reuse[reference:16].• PRISMA-Aligned Curation: The literature review pipeline follows systematic review guidelines, documenting inclusion/exclusion criteria transparently[reference:17]. | The comprehensive data abstraction following Standard Operating Procedures (SOPs) ensures the data is sufficiently well-described to be replicated or combined in new analyses[reference:18]. |
Experimental Protocol: The ECOTOX Systematic Review and Data Curation Pipeline
The reproducibility and fairness of ECOTOX data are grounded in a meticulously documented, multi-stage curation pipeline. The following protocol outlines the key experimental methodology.
Protocol: ECOTOX Literature Search, Review, and Data Curation Pipeline
Objective: To identify, extract, and curate ecologically relevant toxicity data from the scientific literature into a structured, queryable knowledgebase.
Materials:
- Source Literature: Peer-reviewed journals and grey literature (government reports, conference proceedings).
- Bibliographic Databases: Scopus, PubMed, and other subject-specific indices.
- Curation Software: Internal "Unify" database interface for data entry and management.
- Standard Operating Procedures (SOPs): Documents governing each step of the pipeline[reference:19].
Procedure:
- Chemical Verification & Search Strategy Development: For a target chemical, verify its CAS RN. Develop a comprehensive search string using chemical names, synonyms, and related terms.
- Literature Search & Citation Identification: Execute the search strategy across multiple bibliographic databases. Compile all retrieved citations into a reference manager.
- Title/Abstract Screening: Review titles and abstracts against pre-defined Applicability Criteria (Population, Exposure, Comparator, Outcome - PECO)[reference:20]. Exclude irrelevant studies.
- Full-Text Review: Obtain and review the full text of potentially applicable studies. Apply Acceptability Criteria, requiring documented controls and reported toxicity endpoints[reference:21].
- Data Abstraction: For each included study, trained curators extract detailed information into structured fields using the "Unify" interface. Data includes:
- Chemical: Name, CAS RN, purity, formulation, measured/nominal concentration[reference:22].
- Species: Verified scientific name, life stage, source[reference:23].
- Study Design: Test method (e.g., OECD guideline), exposure duration, control type, environmental parameters[reference:24].
- Test Results: Specific effect, endpoint (e.g., LC50, NOEC), statistical significance, units[reference:25].
- Quality Assurance: Extracted data undergoes peer review by a second curator to ensure accuracy and consistency with SOPs.
- Data Maintenance & Release: Curated data is uploaded to the production database. The public website and API are updated quarterly[reference:26].
Validation: The pipeline's reliability is validated through the reproduction of datasets using the independent ECOTOXr R package, which demonstrates high fidelity to manual extractions[reference:27].
Visualizing the Workflow and Principles
Diagram 1: ECOTOX Data Curation Pipeline
Diagram 2: The FAIR Principles Framework
Table 3: Research Reagent Solutions for FAIR Data Curation and Analysis
| Tool / Resource | Function in FAIRification | Relevance to ECOTOX/Field |
|---|---|---|
| ECOTOXr R Package[reference:28] | Provides programmatic, reproducible access to the ECOTOX database. Ensures transparent and consistent data retrieval for meta-analyses. | Key tool for achieving Accessible and Reusable data, enabling scripted workflows that enhance reproducibility. |
| CAS Registry Numbers | Universal, persistent identifiers for chemical substances. | Fundamental for Findability and Interoperability, ensuring unambiguous chemical identification across databases[reference:29]. |
| Controlled Vocabularies & Ontologies (e.g., ECOTOX effect terms, OBO Foundry ontologies) | Standardize terminology for effects, endpoints, and test methods. | Critical for Interoperability, allowing data from different sources to be integrated and queried coherently. |
| Standard Operating Procedures (SOPs) | Documented, step-by-step protocols for literature review and data extraction[reference:30]. | The foundation of ECOTOX's curation pipeline, ensuring consistency, transparency, and Reusability of the data. |
| Persistent Identifier Systems (e.g., DOI for publications) | Provide stable links to source data and metadata. | Supports Findability and provenance tracking, a core aspect of the curation pipeline. |
| Structured Data Formats (e.g., relational databases, CSV with schema) | Organize data in a consistent, machine-readable manner. | Enables Interoperability and efficient data exchange, as seen in the ECOTOX internal database structure. |
The ECOTOX knowledgebase exemplifies a mature, large-scale implementation of FAIR principles within environmental science. By embedding findability through rich metadata, accessibility via open and programmable interfaces, interoperability through standardized identifiers and vocabularies, and reusability via rigorous, systematic curation protocols, ECOTOX transforms dispersed literature into a powerful, trustworthy resource for global risk assessment and research. This case study underscores that adherence to FAIR principles is not an abstract ideal but a practical, achievable framework that significantly amplifies the value and impact of curated scientific data.
This technical guide is framed within a broader thesis investigating the systematic curation of ecotoxicological data to bridge the gap between empirical toxicity testing and modern, mechanistic risk assessment. The central thesis posits that robust, transparent, and well-documented data curation processes are critical for transforming raw, heterogeneous data from sources like the US EPA's ECOTOXicology Knowledgebase (ECOTOX) into reliable, actionable knowledge for regulatory and scientific decision-making [1].
The traditional paradigm of chemical risk assessment, heavily reliant on a limited set of standardized single-species tests, faces significant challenges. These include the vast number of chemicals in commerce, the ethical and practical pressures to reduce vertebrate testing, and the need to assess complex mixture effects and subtle, chronic endpoints such as endocrine disruption [28] [38]. In response, the field is evolving towards New Approach Methodologies (NAMs) and Adverse Outcome Pathway (AOP) frameworks, which require high-quality, curated data on chemical Mode of Action (MoA) for model development, validation, and cross-species extrapolation [28] [1].
The ECOTOX Knowledgebase, as the world's largest curated repository of single-chemical ecotoxicity data, serves as a primary source for such curation efforts [1] [4]. This case study details a replicable methodology for harvesting, curating, and applying ECOTOX data, specifically focusing on deriving MoA classifications and effect concentrations for aquatic species. This process exemplifies the core thesis argument: that meticulous curation is not merely data management but a fundamental research activity that enhances data utility for ecological relevance, supports the grouping of chemicals by biological action, and enables the development of cumulative assessment groups for mixture risk evaluation [28].
Methodology: The Data Curation Pipeline
The curation pipeline follows a systematic, multi-step process aligned with systematic review principles to ensure transparency, objectivity, and reproducibility [1]. The workflow, detailed in the diagram below, integrates source compilation, data harvesting, and multi-tiered curation.
Source Compilation and Data Harvesting
The process begins with compiling a target list of environmentally relevant chemicals. A recent study curated 3,387 compounds, including parent substances, transformation products, and metals, identified from monitoring data, regulatory directives, and scientific literature [28].
Effect concentration data are then harvested from ECOTOX for three key aquatic taxonomic groups aligned with the EU Water Framework Directive's Biological Quality Elements: algae, crustaceans, and fish [28]. ECOTOX provides a comprehensive source, with data compiled from over 53,000 references, covering more than 13,000 species and 12,000 chemicals [4].
Tiered Curation and Quality Assurance
Harvested data undergoes a rigorous two-tiered curation process.
Tier 1: Data Acceptability Screening. Each data point is evaluated against formal ECOTOX and OPP (Office of Pesticide Programs) acceptance criteria [6]. These criteria ensure scientific relevance and quality. The core requirements for a study to be accepted include:
- The study examines single-chemical exposure.
- It reports a biological effect on a live, whole aquatic or terrestrial organism.
- A concurrent exposure concentration/dose and explicit exposure duration are provided.
- The tested species is verified and reported.
- Treatments are compared to an acceptable control group [6]. Studies failing these criteria are excluded from the final curated dataset.
Tier 2: Mode-of-Action Curation. For each chemical, a targeted investigation is conducted to assign a Mode of Action (MoA). This involves searching specialized databases (e.g., EPA MOAtox, PPDB), regulatory documents, and primary literature using the compound name with terms like "mode of action" or "mechanism" [28]. The MoA is categorized according to standardized schemes (e.g., Verhaar scheme) or described specifically, focusing on the molecular initiating event or key physiological target (e.g., "acetylcholinesterase inhibition," "photosystem II inhibitor") [28].
Quantitative Data Output: Curated Results
The output of this pipeline is a structured, FAIR (Findable, Accessible, Interoperable, Reusable) dataset. The tables below summarize the quantitative scope and key findings from a large-scale curation effort [28].
Table 1: Scope of Curated Chemical Dataset by Use Group
| Use Group | Parent Compounds | Transformation Products | Total | Primary Source/Examples |
|---|---|---|---|---|
| Pharmaceutical / Drug of Abuse | 1,162 | 139 | 1,301 | Human & veterinary medicine |
| Pesticide / Biocide | 696 | 204 | 900 | Herbicides, insecticides, fungicides |
| Industrial Chemical | 726 | 19 | 745 | Plasticizers, surfactants, flame retardants |
| Naturally Occurring | 93 | 4 | 97 | Alkaloids, hormones, plant metabolites |
| Metal | 19 | 0 | 19 | Cadmium, copper, zinc, etc. |
| Food Additive | 11 | 0 | 11 | Preservatives, colorants, fragrances |
| Total (Unique) | 2,890 | 374 | 3,387 |
Table 2: Distribution of Major Mode of Action (MoA) Categories
| Mode of Action Category | Approx. % of Classified Compounds | Example Targets/Processes |
|---|---|---|
| Nervous System | ~25% | Acetylcholinesterase, GABA receptor, Sodium channel |
| Endocrine System | ~15% | Estrogen receptor, Androgen receptor, Thyroid axis |
| Photosynthesis Inhibitor | ~10% | Photosystem II (D1 protein), Photosystem I |
| Metabolic/Respiration | ~10% | Mitochondrial complex, Uncoupling agent |
| Cell Membrane/Growth | ~8% | Fatty acid synthesis, Cell division |
| Multiple/Unspecific | ~20% | Narcosis, Oxidative stress, Reactive toxicity |
| Unknown/Unclassified | ~12% | Insufficient mechanistic data available |
Experimental Protocols for Key Analyses
Protocol: Building a Species Sensitivity Distribution (SSD) from Curated Data
SSDs are a key ecological application of curated ECOTOX data, used to derive protective environmental quality benchmarks [38].
1. Data Selection: From the curated dataset, extract all accepted LC50 or EC50 values for a single chemical across multiple species within a defined taxonomic group (e.g., freshwater fish).
2. Data Transformation: Log-transform all effect concentration values (usually to base 10).
3. Distribution Fitting: Rank the transformed values and fit a statistical distribution (e.g., log-normal, log-logistic) using software like R (fitdistrplus package) or the EPA's SSD Tool.
4. Hazard Concentration Derivation: Calculate the HC₅ (Hazard Concentration for 5% of species), which is the concentration estimated to protect 95% of species. The HC₅ is often used as a basis for environmental quality criteria.
5. Uncertainty Analysis: Determine confidence intervals around the HC₅ using bootstrapping or other statistical methods.
Protocol: MoA-Based Chemical Grouping for Mixture Assessment
This protocol uses curated MoA data to identify chemicals for cumulative risk assessment [28].
1. Group Identification: Cluster chemicals from the curated list based on identical or similar curated MoA (e.g., all "Photosystem II inhibitors").
2. Potency Normalization: For each chemical in the group, obtain its Potency Value (e.g., EC50 for a standard test species like Daphnia magna). Calculate a Relative Potency Factor (RPF) by designating the most potent chemical as the index compound (RPF=1) and expressing others as ratios of their EC50s.
3. Mixture Toxicity Prediction: For an environmental mixture, the combined effect can be estimated using the Concentration Addition model: Σ (Ci / EC50i), where Ci is the concentration of chemical i in the mixture. This sum indicates the expected total effect.
Visualization of Conceptual and Workflow Relationships
From Curated Data to Regulatory Application
The following diagram maps the logical pathway from raw data to risk assessment outcomes, illustrating the thesis's core premise on the value of curation.
Table 3: Key Resources for ECOTOX Data Curation and MoA Research
| Tool / Resource Name | Function / Purpose | Key Features / Notes |
|---|---|---|
| US EPA ECOTOX Knowledgebase | Primary source for harvesting single-chemical ecotoxicity test results. | Publicly available; >1M records; advanced search filters for species, endpoint, effect [1] [4]. |
| CompTox Chemicals Dashboard | Chemical identifier resolution, property data, and linkage to ECOTOX. | Provides curated lists, SMILES, InChIKeys, and direct links to ECOTOX search results [4]. |
| EPA ASTER (Assessment Tools for Evaluation of Risk) | QSAR tool for predicting ecological MoA and toxicity. | Can assist in MoA classification for data-poor chemicals [28]. |
| Pesticide Properties Database (PPDB) | Authoritative source for pesticide MoA, chemistry, and toxicity data. | Uses standardized HRAC (herbicide), FRAC (fungicide), IRAC (insecticide) MoA codes. |
| ChEMBL / PubChem BioAssay | Databases of bioactive molecules with mechanistic and target information. | Useful for researching pharmacological MoAs relevant to pharmaceuticals and endocrine disruptors. |
| R Statistical Environment | Data cleaning, statistical analysis (SSD fitting), and visualization. | Essential packages: fitdistrplus, ssdtools, ggplot2, dplyr. |
| Systematic Review Management Software | Managing references and screening processes during large-scale curation. | Tools like Rayyan, Covidence, or DistillerSR streamline title/abstract and full-text review. |
The paradigm in toxicology is decisively shifting from observational, whole-animal studies toward predictive, mechanistic science based on New Approach Methodologies (NAMs). This shift, driven by the need to assess thousands of chemicals efficiently while reducing animal testing, relies on two pillars: in silico models like Quantitative Structure-Activity Relationships (QSAR) and conceptual frameworks like the Adverse Outcome Pathway (AOP) [39]. However, the development, validation, and regulatory acceptance of these NAMs are critically dependent on access to high-quality, curated in vivo toxicity data [1].
The ECOTOXicology Knowledgebase (ECOTOX) serves as this foundational empirical resource. As the world's largest curated compilation of single-chemical ecotoxicity data, it provides the essential link between traditional toxicology and next-generation methodologies [1]. ECOTOX supports the entire NAM development pipeline: its data is indispensable for training and validating QSAR models, anchoring and quantifying AOPs, and enabling the in vitro to in vivo extrapolations (IVIVE) necessary for risk assessment [4]. This technical guide details how systematically curated in vivo data from ECOTOX is leveraged to advance computational and mechanistic toxicology within the broader context of modern data curation research.
Table: The Scope and Role of the ECOTOX Knowledgebase
| Metric | Description | Role in Supporting NAMs |
|---|---|---|
| Test Records | Over 1 million curated results [1] [4] | Provides a large-scale training and validation dataset for computational models (e.g., QSAR). |
| Chemical Coverage | Over 12,000 single chemical stressors [1] [4] | Enables exploration of chemical space and structure-activity relationships across diverse classes. |
| Species Coverage | Over 13,000 aquatic and terrestrial species [4] | Supports cross-species extrapolation and understanding of taxonomic susceptibility. |
| References | Data compiled from over 53,000 sources [4] | Ensures evidence is based on a comprehensive, transparent literature foundation. |
Systematic Data Curation: The ECOTOX Pipeline for Reliable Evidence
The utility of ECOTOX data for sensitive applications like QSAR and AOP development hinges on the rigor and transparency of its curation process. The pipeline follows principles aligned with systematic review methodologies, ensuring data integrity and fitness-for-purpose [1].
The ECOTOX curation process is a multi-stage funnel designed to identify, evaluate, and extract relevant ecotoxicity data with maximal consistency [1].
- Literature Search & Screening: Comprehensive searches of open and "grey" literature are conducted using chemical-specific terms. Titles, abstracts, and finally full texts are screened against pre-defined eligibility criteria (e.g., single chemical tested, ecologically relevant species, reported exposure concentration and duration) [1].
- Data Extraction & Curation: From accepted studies, trained reviewers extract detailed information into a controlled vocabulary system. This includes chemical identity, species taxonomy, meticulous test condition parameters (exposure route, duration, media), and toxicological endpoints (mortality, growth, reproduction). A key feature is the extraction of the exact quantitative result (e.g., LC50, NOEC) and its associated value, unit, and statistical information [1].
- Quality Assurance & Publication: Extracted data undergoes quality control checks before being added to the public knowledgebase, which is updated quarterly. The process ensures data are Findable, Accessible, Interoperable, and Reusable (FAIR), directly supporting computational reuse [1].
Diagram: ECOTOX Systematic Data Curation Pipeline. The workflow transforms literature into FAIR data suitable for NAM development.
Experimental Protocol: Implementing a Systematic Review for Data Curation
The following protocol, based on ECOTOX's standard operating procedures [1], can be adapted for targeted data curation to support specific NAM projects:
- Protocol Development: Define the specific chemical classes and apical outcomes (e.g., fish acute mortality, invertebrate reproduction) of interest. Establish explicit inclusion/exclusion criteria for test organisms, study design, and data reporting.
- Search Strategy: Develop a structured search string using chemical names and CAS numbers. Execute searches across multiple bibliographic databases (e.g., PubMed, Web of Science, Scopus) and regulatory or grey literature sources.
- Study Screening: Use a two-phase screening process (title/abstract, then full-text) with two independent reviewers to minimize bias. Resolve conflicts through consensus or a third reviewer.
- Data Extraction: Using a pre-piloted form, extract key variables: chemical verification data, species Latin name and life stage, test conditions (duration, temperature, media), endpoint type, quantitative result with unit and measure of dispersion (e.g., standard error), and study quality indicators (e.g., solvent controls, compliance with test guidelines).
- Data Management & Curation: Apply controlled vocabularies (e.g., from ECOTOX or EPA's CompTox Dashboard) for chemicals and endpoints. Perform unit conversions to a standard system (e.g., molarity). Conduct plausibility checks on reported values before integrating the data into the analysis-ready dataset.
Powering Predictive Models: In Vivo Data for QSAR Development
Quantitative Structure-Activity Relationship (QSAR) models are essential in silico NAMs for predicting toxicity from chemical structure. Their reliability is a direct function of the quality and relevance of the in vivo data used to build them.
The Role of Curated Data in QSAR Model Development
Curated in vivo data from ECOTOX addresses critical needs in the QSAR modeling pipeline [1] [40]:
- Training Data: Provides the experimental toxicity values (e.g., LC50, EC10) that serve as the dependent variable in model training.
- Chemical Space Definition: The diversity of over 12,000 chemicals helps define the "applicability domain" of a model—the range of structures for which it can make reliable predictions.
- External Validation: A robust, independent dataset from ECOTOX is used to test a model's predictive performance on new chemicals, which is a cornerstone of OECD validation principles.
- Mechanistic Interpretation: Data for specific, mechanistically linked endpoints (e.g., acetylcholinesterase inhibition leading to mortality) allows development of more interpretable, mechanism-based QSARs aligned with AOPs [41].
Experimental Protocol: Building an AOP-Informed QSAR Model
This protocol outlines the integration of curated in vivo data for developing a QSAR model targeting a Molecular Initiating Event (MIE) within an AOP [41] [40].
- Endpoint Selection & Data Acquisition: Select a protein target related to an MIE (e.g., binding to the aryl hydrocarbon receptor). Curate in vitro bioactivity data (e.g., IC50 values from ChEMBL) for this target, converting it to a binary (active/inactive) or quantitative (pChEMBL) format. In parallel, curate relevant in vivo apical endpoint data from ECOTOX for chemicals with bioactivity data [41].
- Descriptor Calculation & Data Curation: Calculate molecular descriptors (e.g., topological, electronic, geometrical) and fingerprints for all chemicals. Clean the dataset by removing duplicates and salts, and standardizing structures.
- Model Training & Validation: Split the in vitro bioactivity data into training and test sets. Employ machine learning algorithms (e.g., Random Forest, Support Vector Machines) to build a classification or regression model predicting bioactivity from molecular descriptors. Optimize hyperparameters using cross-validation. Validate the model's external predictivity using the held-out test set [41].
- Linking In Silico Prediction to In Vivo Outcome: Apply the validated QSAR model to screen new chemicals for MIE activity. For chemicals predicted as active, use the parallel in vivo dataset from ECOTOX to investigate empirical correlations between predicted MIE potency and observed apical toxicity, thereby strengthening the AOP's quantitative linkages [41].
Anchoring Mechanistic Frameworks: Data for AOP Development and Quantification
The Adverse Outcome Pathway framework organizes knowledge into a sequence of causally linked events from a Molecular Initiating Event to an Adverse Outcome. In vivo data is crucial for building confidence in these pathways.
Using Empirical Data in the AOP Lifecycle
- Identification of Key Events (KEs): Analysis of curated in vivo studies can reveal consistent, intermediate biological effects (e.g., histopathological changes, altered enzyme activity) that serve as candidate KEs between an MIE and an AO [39].
- Weight-of-Evidence Assessment: ECOTOX data provides evidence to assess the essentiality of a proposed KE. For example, the consistent observation of liver necrosis preceding population decline in fish across multiple chemical classes supports its role as a KE in a relevant AOP.
- Quantitative AOP (qAOP) Development: This is the most data-intensive stage. Curated dose-response and time-course data for each KE are used to establish quantitative relationships between KEs (e.g., predictive regression models), transforming a qualitative pathway into a predictive network [39]. ECOTOX's standardized data is ideal for this meta-analysis.
Diagram: Integration of QSAR Predictions with the AOP Framework. QSAR models predict the MIE, while curated in vivo data from ECOTOX validates the connection to downstream adverse outcomes.
Experimental Protocol: Systematic Evidence Mapping for AOP Development
A systematic, evidence-based approach strengthens AOP development [42].
- Framing the AOP Question: Formulate a PECO (Population, Exposure, Comparator, Outcome) statement. Example: "In freshwater fish (P), does exposure to chemicals that bind to the estrogen receptor (E), compared to unexposed controls (C), lead to population-level reproductive failure (O)?" [42]
- Evidence Collection: Conduct systematic literature searches for each proposed KE (e.g., vitellogenin induction, altered gonad histology, reduced fecundity). ECOTOX can be a primary source for apical outcome data (O).
- Evidence Evaluation & Synthesis: For each retrieved study, assess the risk of bias (e.g., study design, reporting quality). Extract data on dose-response, temporal sequence, and incidence. Synthesize evidence to evaluate the strength and consistency of support for each Key Event Relationship (KER).
- AOP Assembly & Confidence Grading: Assemble the supported KEs into a pathway. Grade confidence in the AOP (Low, Moderate, High) based on the weight of evidence for biological plausibility, empirical support, and essentiality of the KEs [42].
Bridging the Gap: In Vivo Data for In Vitro to In Vivo Extrapolation (IVIVE)
IVIVE is a critical modeling process that converts an active concentration from an in vitro assay into an equivalent external dose expected to cause an effect in vivo, enabling the use of high-throughput screening data in risk assessment [43].
The Central Role of Toxicokinetics and Reverse Dosimetry
The core challenge of IVIVE is accounting for the physiological processes of Absorption, Distribution, Metabolism, and Excretion (ADME) that a chemical undergoes in a whole organism. This is addressed through Toxicokinetic (TK) modeling and reverse dosimetry [43].
- Point of Departure (POD): An active concentration (e.g., AC50) is identified from an in vitro assay targeting an MIE or KE.
- Reverse Dosimetry: A TK model (from simple one-compartment to complex PBPK models) is run "in reverse." The in vitro POD is set as a target steady-state blood or tissue concentration, and the model calculates the daily external intake dose required to achieve it.
- Comparison to In Vivo Data for Validation: The predicted external dose from IVIVE is compared to actual effect doses from traditional in vivo studies (e.g., from ECOTOX). Close agreement increases confidence in the IVIVE approach and the in vitro assay as a surrogate for that particular endpoint [43] [44].
Diagram: IVIVE Workflow for Translating In Vitro Data to In Vivo Doses. Curated in vivo data is essential for validating the predictions generated through reverse dosimetry.
Table: Key Steps in a NAM Data Integration Workflow
| Step | Action | Tools / Data Sources | Output |
|---|---|---|---|
| 1. Define Scope | Identify chemical space & apical outcome of regulatory concern. | Regulatory mandates, AOP Wiki. | Defined problem statement. |
| 2. Gather In Vivo Evidence | Conduct systematic review & curate existing in vivo toxicity data. | ECOTOX Knowledgebase, published literature. | Curated dataset of apical outcomes. |
| 3. Develop In Silico Model | Build QSAR model for MIE or early KE using curated in vitro data. | ChEMBL, ToxCast data, modeling software. | Validated predictive model. |
| 4. Perform IVIVE | Use TK modeling to convert in vitro bioactivity to predicted in vivo dose. | PBPK/IVIVE platforms (e.g., EPA's httk). | Predicted point of departure (POD). |
| 5. Validate & Integrate | Compare IVIVE predictions to curated in vivo data from Step 2. | Statistical analysis tools. | Validated, integrated testing strategy for decision-making. |
Table: Key Research Reagent Solutions for NAM Development
| Tool / Resource | Function in NAM Development | Key Features / Examples |
|---|---|---|
| ECOTOX Knowledgebase | Primary source of curated in vivo ecotoxicity data for model training and validation. | >1M test results; advanced querying; links to chemical databases [1] [4]. |
| EPA CompTox Chemicals Dashboard | Provides curated chemical structures, properties, and identifiers for QSAR descriptor calculation. | Integrated with ECOTOX and ToxCast data; supports chemical space analysis [4]. |
| AOP-Wiki (OECD) | Central repository for collaborative AOP development and knowledge organization. | Houses ~400 AOPs; framework for structuring mechanistic data [41] [39]. |
| ChEMBL Database | Source of curated in vitro bioactivity data for MIE targets (e.g., receptor binding, enzyme inhibition). | Essential for developing MIE-targeted QSAR models [41]. |
| ToxCast/Tox21 Data | High-throughput screening (HTS) data for thousands of chemicals across hundreds of assay endpoints. | Used for bioactivity profiling and as input for IVIVE [43]. |
| IVIVE/PBPK Modeling Platforms | Software tools to perform reverse dosimetry and extrapolate from in vitro to in vivo doses. | Examples include EPA's "httk" R package and commercial simulators (e.g., GastroPlus, Simcyp) [43]. |
| Controlled Vocabulary Systems | Standardized terms for chemicals, species, and endpoints to ensure data interoperability. | Critical for merging data from different sources (ECOTOX, ChEMBL, ToxCast) into unified datasets. |
The advancement of NAMs is not about discarding in vivo data but about leveraging it more intelligently. The ECOTOX Knowledgebase exemplifies how rigorous, systematic curation of traditional studies creates an indispensable asset for the future of predictive toxicology. By providing the empirical anchor for QSAR model validation, the evidential foundation for AOP development, and the benchmark for IVIVE predictions, high-quality in vivo data transforms from an endpoint into a catalyst. This integrated, data-centric approach, framed within robust curation research, enables a more efficient, mechanistic, and ultimately protective paradigm for chemical safety assessment.
The data curation processes underlying ecotoxicological knowledgebases are foundational to robust chemical risk assessment and environmental research. Within the context of a broader thesis on data curation, this analysis examines two distinct paradigms: the comprehensive, global ECOTOX database maintained by the U.S. Environmental Protection Agency (EPA) and regionalized databases exemplified by California’s CalEcotox [45] [6]. While both serve to bridge primary literature and risk assessment conclusions, their design philosophies, curation protocols, and intended applications differ significantly. Understanding these differences is critical for researchers and drug development professionals to select the appropriate tool for targeted assessments, whether for broad ecological screening or region-specific conservation planning.
Core Database Architecture and Scope
The fundamental design and scope of ECOTOX and CalEcotox dictate their utility. The following table summarizes their contrasting architectures.
Table 1: Comparative Scope and Design of ECOTOX and CalEcotox
| Feature | ECOTOX (U.S. EPA) | CalEcotox (California) |
|---|---|---|
| Primary Objective | Support national & international chemical risk assessments; serve as a comprehensive search engine for ecological effects data [6]. | Support ecotoxicological risk assessments specific to California’s wildlife and habitats [45]. |
| Spatial Scope | Global. Data is collected from worldwide literature without geographic restriction [6]. | Regional. Focuses exclusively on species known to occur in California, though may include data from studies conducted elsewhere for those species [45]. |
| Taxonomic & Habitat Focus | Broad. Includes aquatic and terrestrial plants and animals from all global habitats [6]. | Targeted. Primarily terrestrial and semi-terrestrial vertebrates (mammals, birds, reptiles, amphibians); one fish species added recently [45]. |
| Data Types | Primarily toxicological dose-response data (e.g., LC50, EC50, NOEC). Includes detailed test condition metadata [46]. | Integrated data: Combines species-specific exposure factors (body weight, ingestion rates, home range) with toxicological endpoints and bioaccumulation data [45]. |
| Governance & Users | U.S. EPA Office of Research and Development. Used by EPA’s Office of Pesticide Programs under agreement with U.S. Fish and Wildlife Service [6]. | California Office of Environmental Health Hazard Assessment (OEHHA). Designed for use by state and federal agencies assessing risks in California [45]. |
ECOTOX is structured as a relational database with interconnected tables for tests, results, species, and chemicals, allowing for complex queries [46]. Its schema is designed to capture the vast heterogeneity of global ecotoxicology studies. In contrast, CalEcotox is a species-driven relational database that prioritizes the synthesis of physiological, ecological, and toxicological parameters for a curated list of regionally relevant species [45].
Data Curation Methodology and Quality Assurance
The processes for identifying, extracting, and validating data are where the core philosophical differences between comprehensive and regional databases become most apparent. These methodologies directly impact the fitness of data for different assessment types.
Table 2: Data Curation and Quality Assurance Protocols
| Curation Phase | ECOTOX Protocols | CalEcotox Protocols |
|---|---|---|
| Literature Search & Acquisition | Systematic searches conducted by EPA’s Mid-Continental Ecology Division for pesticides in Registration Review [6]. Relies on the public ECOTOX interface for other chemicals. | Two-tiered approach: 1) Electronic database searches (e.g., Biosis Previews, Zoological Record); 2) Review of primary & secondary sources for older literature. Original searches completed in 1999, updated in 2018 [45]. |
| Study Acceptance Criteria | Studies must meet minimum criteria: single chemical exposure, effect on whole live organism, reported concentration/dose, explicit exposure duration [6]. OPP applies additional screens (e.g., English language, full article, calculated endpoint) [6]. | Data sourced from peer-reviewed journals, theses, government reports. Only species-specific empirical data from literature is entered; parameters with no published information remain as data gaps [45]. |
| Data Entry & Standardization | Data coded into structured tables (tests, results, doses) with extensive metadata [46]. | Data entered as datasets linked to citation information. Each dataset includes values and descriptors about study design [45]. |
| Quality Assurance / Aggregation | Provides raw data. Third-party tools like Standartox have been developed to standardize ECOTOX data, filter by endpoint/quality, and calculate aggregate values (geometric mean) to reduce variability [47]. | No allometric estimates or derived data. Provides only captured empirical values, presenting data variability directly to the user [45]. |
The experimental protocol for curating data into CalEcotox involves a defined multi-step workflow [45]:
- Species Selection: Species are chosen based on occurrence in California, utility as indicator/surrogate species, trophic level, and legal status (threatened/endangered).
- Comprehensive Literature Retrieval: For each species, structured searches are executed across multiple bibliographic databases (e.g., Aquatic Sciences Abstracts, Wildlife Worldwide, Zoological Record).
- Data Extraction and Gap Analysis: Relevant exposure factors and toxicological endpoints are extracted from acceptable sources. Missing parameters for a species are explicitly noted as gaps.
- Database Population: Extracted data is entered into the relational database with intact links to the original source citation.
For ECOTOX, the evaluation guidelines for the Office of Pesticide Programs detail a protocol for screening and reviewing open literature [6]:
- ECOTOX Search & Initial Categorization: ORD/MED performs searches and categorizes papers as: Accepted; Accepted by ECOTOX but not OPP; Rejected; or "Other."
- Risk Assessor Screening: The risk assessor applies OPP acceptability criteria (e.g., toxicant relevance, primary source, acceptable control) to identify useful papers.
- Study Review and Classification: Accepted studies are reviewed for quality and classified based on their utility (e.g., for quantitative endpoint derivation, qualitative support, or mode of action information).
- Integration into Assessment: Data from accepted studies are used to refine toxicity endpoints, support existing data, or identify hazards.
A key development in the ecosystem of ecotoxicological data is the emergence of tools like Standartox, which builds directly upon the curated data in ECOTOX [47]. Standartox implements a post-curation processing workflow that addresses data variability:
- Data Ingestion: Incorporates the quarterly updates from the ECOTOX database.
- Filtering and Harmonization: Restricts data to common endpoints (EC50, NOEC, etc.), standardizes units, and allows filtering by effect group, chemical role, organism habitat, and geographic distribution.
- Aggregation: For multiple test results for the same chemical-organism combination, it calculates aggregate statistics (minimum, maximum, geometric mean) to produce a single, standardized toxicity value, reducing selection bias and uncertainty in subsequent analyses [47].
The following diagram illustrates the overarching data curation and application workflow connecting primary research, global databases, regional tools, and analytical applications.
Diagram 1: Ecotox Data Curation and Application Workflow (84 characters)
Selecting and effectively utilizing these databases requires a suite of complementary tools and resources. The following table details key components of a research toolkit for conducting targeted ecotoxicological assessments.
Table 3: Research Reagent Solutions for Ecotoxicological Assessments
| Tool / Resource | Function | Relevance to Assessment Type |
|---|---|---|
| ECOTOX Web Interface / API | Primary portal for querying the global EPA database by chemical, species, or endpoint [6]. | Essential for broad-scope hazard identification and literature review for chemicals with widespread use. |
ECOTOXr R Package |
Allows direct querying of a local copy of the full ECOTOX relational database, enabling complex, reproducible analyses beyond web interface limitations [46]. | Critical for researchers developing automated workflows, performing meta-analyses, or needing to join specific test condition data with results. |
| Standartox Web App & R Package | Provides cleaned, filtered, and aggregated values from ECOTOX. Calculates geometric means for chemical-species pairs to reduce variability [47]. | Highly useful for deriving stable benchmark values (e.g., for Species Sensitivity Distributions) and for comparative screening of chemicals. |
| CalEcotox Database | Integrated source of California-specific species biology, exposure parameters, and toxicity data [45]. | Indispensable for ecological risk assessments mandated under California law or focused on California’s unique ecosystems and protected species. |
| EPA Chemicals Dashboard | Provides complementary data on chemical properties, environmental fate, and human health toxicity, aiding in the interpretation of ecotoxicology data [35]. | Useful for understanding chemical behavior (e.g., bioaccumulation potential, persistence) which informs exposure and hazard in assessments. |
| Knowledge Extraction & Text-Mining Software (e.g., IRIS) | Semi-automates the extraction of toxicological knowledge and relationships from vast scientific literature [48]. | Supports the data curation process itself, helping to identify new data for inclusion in databases or to map mechanisms of action. |
Application in Targeted Assessments: Strategic Selection
The choice between ECOTOX and a regional database is not mutually exclusive but should be guided by the assessment’s specific problem formulation.
Use ECOTOX for: Broad-Scale Hazard Characterization, Chemical Prioritization, and Global or National Regulatory Assessments. Its strength is in providing the widest possible view of a chemical’s tested effects across the tree of life, which is necessary for initial screening and for assessments covering large geographic areas [6]. When combined with a post-processing tool like Standartox, it offers a powerful method to generate standardized toxicity values for use in models like Species Sensitivity Distributions (SSDs) [47].
Use CalEcotox (or analogous regional DBs) for: Region-Specific Risk Characterization, Assessments for Listed/Threatened Species, and Refined Exposure Estimation. Its integrated design is its greatest asset. For a risk assessment on a California vernal pool ecosystem, CalEcotox provides pre-compiled, species-specific exposure factors (e.g., foraging distance, dietary composition) for local species that are not available in ECOTOX. This directly supports a higher-tier, realistic exposure assessment without resorting to generic models [45].
Within the thesis of ECOTOX knowledgebase data curation, this analysis highlights that the curation objective dictates the product. ECOTOX’s curation aims for maximal breadth and replicability of toxicological test conditions, serving as a foundational data warehouse. CalEcotox’s curation aims for practical synthesis of disparate data types (exposure + effect) for a defined ecological context. The future of ecotoxicological data curation lies in enhancing interoperability between these models. This could involve the development of regional "overlays" that filter and contextualize global data, or the adoption of standardized data formats that allow regional databases to seamlessly integrate aggregated, quality-controlled data from tools like Standartox. For the researcher, the most robust targeted assessment will strategically leverage the global breadth of ECOTOX and the contextual depth of regional databases, using the growing toolkit of software packages to bridge the gap between raw data and actionable scientific conclusions.
Conclusion
The ECOTOX Knowledgebase's rigorous, systematic curation process is foundational to modern ecological risk assessment and chemical safety science. By transforming disparate study data into a standardized, accessible, and quality-controlled resource, it fulfills critical regulatory mandates and accelerates research. The pipeline's alignment with systematic review principles and FAIR data standards ensures its reliability for diverse applications, from setting water quality criteria to validating computational toxicology models. Future developments will likely focus on enhancing interoperability with other 'omics' databases, further automating the curation pipeline, and expanding its role in supporting the global transition to animal-free New Approach Methodologies. For researchers and drug development professionals, mastering the use of this curated knowledgebase is essential for efficient, credible, and cutting-edge environmental health research.
References
- 1. The ECOTOXicology Knowledgebase: A Curated Database of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9408435/]
- 2. High-throughput screening paradigms in ecotoxicity testing [https://www.sciencedirect.com/science/article/abs/pii/S0045653522024225]
- 3. Proposal of critical appraisal tools for the evaluation of ... - EFSA [https://www.efsa.europa.eu/en/supporting/pub/en-7787]
- 4. Ecotoxicology (ECOTOX) Knowledgebase Resource Hub [https://www.epa.gov/comptox-tools/ecotoxicology-ecotox-knowledgebase-resource-hub]
- 5. ECOTOXr: An R package for reproducible and transparent ... [https://www.sciencedirect.com/science/article/pii/S0045653524019751]
- 6. Evaluation Guidelines for Ecological Toxicity Data in the ... [https://www.epa.gov/pesticide-science-and-assessing-pesticide-risks/evaluation-guidelines-ecological-toxicity-data-open]
- 7. A Framework for Ecotoxicity Testing in the 21st Century [https://www.mdpi.com/2076-3417/9/3/428]
- 8. EPA Releases Proposal to Increase Efficiency, Better ... [https://www.epa.gov/newsreleases/epa-releases-proposal-increase-efficiency-better-protect-health-and-environment]
- 9. Collecting, Evaluating, and Communicating Ecotoxicology Data... [https://www.slideshare.net/slideshow/collecting-evaluating-and-communicating-ecotoxicology-data-ecotox-knowledgebase/277681669]
- 10. cfpub.epa.gov/ ecotox [https://cfpub.epa.gov/ecotox/]
- 11. CompTox Chemicals Dashboard [https://www.epa.gov/comptox-tools/comptox-chemicals-dashboard]
- 12. Ecotoxicity characterization of chemicals: Global ... [https://www.sciencedirect.com/science/article/pii/S0045653522033008]
- 13. Grey Literature - Searching for Systematic Reviews ... [https://libguides.kcl.ac.uk/systematicreview/greylit]
- 14. A Guide to Evidence Synthesis: 3. Select Grey Literature ... [https://guides.library.cornell.edu/evidence-synthesis/grey-literature]
- 15. Ecological Soil Screening Level [https://www.epa.gov/chemical-research/ecological-soil-screening-level]
- 16. Controlled Vocabulary [https://www.librarianshipstudies.com/2020/03/controlled-vocabulary.html]
- 17. Keywords and Controlled Vocabulary - Tips for Searching ... [https://guides.library.illinois.edu/c.php?g=1191165&p=8712542]
- 18. Text has enhanced contrast | ACT Rule | WAI [https://www.w3.org/WAI/standards-guidelines/act/rules/09o5cg/]
- 19. Text color must provide high contrast with its background [https://docs.acquia.com/web-governance/text-color-must-provide-high-contrast-its-background]
- 20. A Curated Database of Ecologically Relevant Toxicity Tests to ... [https://academic.oup.com/etc/article/41/6/1520/7730623]
- 21. A benchmark dataset for machine learning in ecotoxicology [https://www.nature.com/articles/s41597-023-02612-2]
- 22. Downloadable Computational Toxicology Data [https://www.epa.gov/comptox-tools/downloadable-computational-toxicology-data]
- 23. Computational Toxicology and Exposure APIs [https://www.epa.gov/comptox-tools/computational-toxicology-and-exposure-apis]
- 24. Current Status and Planned Updates of TRI Tools [https://www.epa.gov/toxics-release-inventory-tri-program/current-status-and-planned-updates-tri-tools]
- 25. Updating statistical practice in ecotoxicology: reflections ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12047014/]
- 26. Ecotox REACH 2025 Conference: Key Takeaways [https://www.complianceandrisks.com/cr-news/ecotox-reach-2025-conference-key-takeaways-on-reach-2-0-clp-revision-and-pfas-restrictions/]
- 27. Unlocking the future of environmental safety: a framework ... [https://www.sciencedirect.com/science/article/pii/S3050620425000235]
- 28. Curated mode-of-action data and effect concentrations for ... [https://www.nature.com/articles/s41597-023-02904-7]
- 29. Predicting the Aquatic Toxicity of Pharmaceutical and ... [https://www.mdpi.com/2076-3417/15/3/1246]
- 30. EPA Science Training Webinar: ECOTOX Knowledgebase [https://ncceh.ca/events/upcoming-events/epa-science-training-webinar-ecotox-knowledgebase]
- 31. Download and Extract Data from US EPA's ECOTOX Database [https://pepijn-devries.github.io/ECOTOXr/]
- 32. Towards Rational Pesticide Design with Graph Machine ... [https://arxiv.org/html/2509.18703]
- 33. ECOTOXicology Knowledgebase-status of curation of PFAS ... [https://epa.figshare.com/articles/presentation/ECOTOXicology_Knowledgebase-status_of_curation_of_PFAS_data/29420930]
- 34. How To Use Expert Judgment In Project Management [2025] [https://asana.com/resources/expert-judgment]
- 35. Chemicals Dashboard Help: Basic Search [https://www.epa.gov/comptox-tools/chemicals-dashboard-help-basic-search]
- 36. ToxPi*GIS Toolkit: creating, viewing, and sharing ... [https://www.nature.com/articles/s41370-022-00433-w]
- 37. Computational Toxicology and Exposure Online Resources [https://www.epa.gov/comptox-tools]
- 38. An ecological perspective in aquatic ecotoxicology [https://www.sciencedirect.com/science/article/abs/pii/S1439179107001041]
- 39. Adverse outcome pathways – development and potential regulatory... [https://www.redalyc.org/journal/5705/570569953002/html/]
- 40. A Review of Quantitative Structure-Activity Relationship ... [https://pubmed.ncbi.nlm.nih.gov/41012420/]
- 41. Quantitative structure–activity relationships of chemical ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-024-00917-x]
- 42. Applying Evidence-Based Methods to the Development and Use of... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9394185/]
- 43. In vitro to in vivo extrapolation for high throughput prioritization ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7393693/]
- 44. In vitro to in vivo extrapolation to support the development ... [https://pubs.rsc.org/en/content/articlelanding/2022/nr/d2nr00664b]
- 45. CalEcotox – a California Wildlife Biology, Exposure Factor ... [https://ecotox.oehha.ca.gov/]
- 46. ECOTOX database schema [https://cran.r-project.org/web/packages/ECOTOXr/vignettes/ecotox-schema.html]
- 47. Standartox: Standardizing Toxicity Data [https://www.mdpi.com/2306-5729/5/2/46]
- 48. Prior Knowledge for Predictive Modeling: The Case of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9472271/]