Research Articles
Mastering ECOTOX: A Step-by-Step Guide for Researchers to Extract Critical Ecotoxicology Data
This comprehensive tutorial provides scientific researchers, toxicologists, and drug development professionals with essential strategies for effectively navigating the US EPA's ECOTOXicology Knowledgebase.
Computational Tools and Bioinformatics Strategies for Defining the Taxonomic Domain of Applicability of Adverse Outcome Pathways
This article provides a comprehensive guide for researchers and drug development professionals on the critical task of defining the taxonomic domain of applicability (tDOA) for Key Event Relationships (KERs) within...
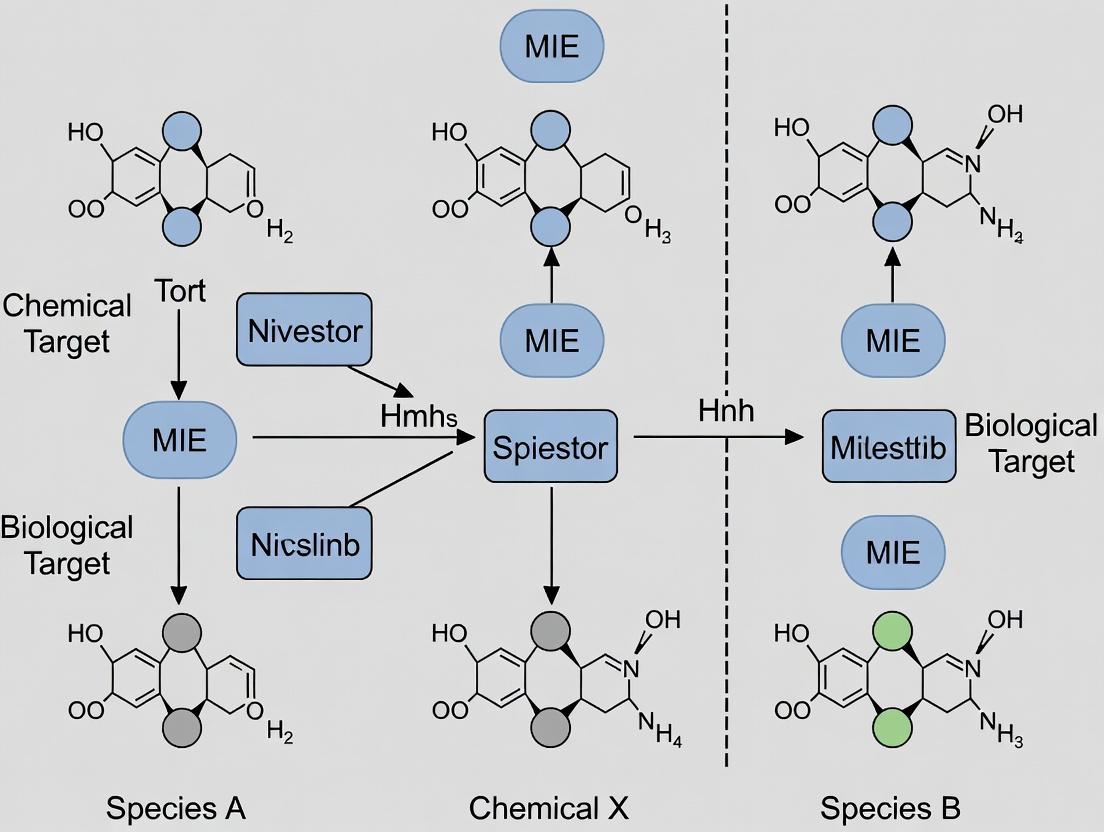
Unlocking Cross-Species Toxicity Predictions: The Critical Role of Molecular Initiating Event Conservation
This article provides a comprehensive analysis of Molecular Initiating Event (MIE) conservation across species, a cornerstone for modern predictive toxicology and chemical safety assessment.
Taxonomic Domains Decoded: A Multidimensional Guide for Biomedical Research and Drug Discovery
This guide provides a comprehensive framework for understanding 'domains' across biological, clinical, and structural contexts, tailored for biomedical researchers and drug development professionals.
Decoding Taxonomic Applicability in AOPs: A Practical Guide for Cross-Species Prediction in Biomedical Research
This article provides a comprehensive guide to the Taxonomic Domain of Applicability (tDOA) in Adverse Outcome Pathways (AOPs), a critical concept for researchers and drug development professionals using these frameworks...
Assessing Species Susceptibility in Adverse Outcome Pathways: A Framework for Predictive Toxicology & Safer Drug Development
This article provides a comprehensive guide for researchers and drug development professionals on evaluating species-specific susceptibility within the Adverse Outcome Pathway (AOP) framework.
AOP-Driven Cross-Species Extrapolation: A Mechanistic Framework for Predictive Toxicology and Drug Safety
This article provides a comprehensive overview of the Adverse Outcome Pathway (AOP) framework as a transformative tool for cross-species extrapolation in toxicology and drug development.
Decoding Taxonomic Applicability in Adverse Outcome Pathways (AOPs): A FAIR Framework for Next-Generation Risk Assessment
This article provides a comprehensive guide for researchers and drug development professionals on the critical role of taxonomic applicability within the Adverse Outcome Pathway (AOP) framework.
Data Quality Assessment Tools for Ecotoxicology: A Comparative Guide for Researchers and Drug Development Professionals
High-quality data is the cornerstone of reliable ecological risk assessment and regulatory decision-making in ecotoxicology.
Ensuring Reliability in Environmental Risk Assessment: A Comprehensive Guide to Interlaboratory Comparison of Ecotoxicity Tests
This article provides a detailed guide to interlaboratory comparison (ILC) studies for ecotoxicity testing, designed for researchers and regulatory professionals.